10-K: Annual report pursuant to Section 13 and 15(d)
Published on August 13, 2013
Table of Contents
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
[X] ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF
THE SECURITIES EXCHANGE ACT OF 1934
For the fiscal year ended June 30, 2013
Commission file number: 001-15317
RESMED INC.
(Exact name of registrant as specified in its charter)
DELAWARE
(State or other jurisdiction of incorporation or organization)
98-0152841
(IRS Employer Identification No.)
9001 Spectrum Center Blvd.
San Diego, CA 92123
United States of America
(Address of principal executive offices)
(858) 836-5000
(Registrants telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
TITLE OF EACH CLASS
Common Stock, $0.004 Par Value
Name of each exchange upon which registered
New York Stock Exchange
Securities registered pursuant to Section 12(g) of the Act
None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes [ x ] No [ ]
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Act. Yes [ ] No [ x ]
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes [ x ] No [ ]
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes [ x ] No [ ]
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulations S-K (§ 229.405 of this Chapter) is not contained herein, and will not be contained, to the best of registrants knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K [ ]
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer or a smaller reporting company. See the definitions of large accelerated filer, accelerated filer and smaller reporting company in Rule 12b-2 of the Exchange Act.
Large accelerated filer [ x ] Accelerated filer [ ] Non-accelerated filer [ ] Smaller reporting company [ ]
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes [ ] No [ x ]
The aggregate market value of the voting and non-voting common equity held by non-affiliates of registrant as of December 31, 2012 (the last business day of the registrants most recently completed second fiscal quarter), computed by reference to the closing sale price of such stock on the New York Stock Exchange, was $5,886,280,490. All directors, executive officers, and 10% stockholders of registrant are considered affiliates.
At August 5, 2013, registrant had 142,054,277 shares of Common Stock, $0.004 par value, issued and outstanding. This number excludes 32,026,013 shares held by the registrant as treasury shares.
Portions of the registrants definitive Proxy Statement to be delivered to shareholders in connection with the registrants 2013 Annual Meeting of Stockholders, to be filed subsequent to the date hereof, are incorporated by reference into Part III of this report.
Table of Contents
As used in this 10-K, the terms we, us, our and the Company refer to ResMed Inc., a Delaware corporation, and its subsidiaries, on a consolidated basis, unless otherwise stated.
- 1 -
Table of Contents
PART I
Cautionary Note Regarding Forward-Looking Statements
This report contains certain forward-looking statements and information that are based on the beliefs of our management as well as estimates and assumptions made by, and information currently available to our management. All statements other than statements regarding historical facts are forward-looking statements. The words believe, expect, anticipate, intend, seek, will, will continue, estimate, plan, future and other similar expressions generally identify forward-looking statements, including, in particular, statements regarding the development and approval of new products and product applications, market expansion, pending litigation, and the development of new markets for our products, such as cardiovascular and stroke markets. These forward-looking statements are made under the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. You are cautioned not to place undue reliance on these forward-looking statements each of which applies only as of the date of this report. Such forward-looking statements reflect the views of our management at the time such statements are made and are subject to a number of risks, uncertainties, estimates and assumptions, including, without limitation, and in addition to those identified in the text surrounding such statements, those identified in Item 1A Risk Factors and elsewhere in this report.
In addition, important factors to consider in evaluating such forward-looking statements include changes or developments in social, economic, market, legal or regulatory circumstances, changes in our business or growth strategy or an inability to execute our strategy due to changes in our industry or the economy generally, the emergence of new or growing competitors, the actions or omissions of third parties, including suppliers, customers, competitors and governmental authorities, and various other factors subject to risks and uncertainties which could cause actual results to materially differ from those projected or implied in the forward-looking statements. Should any one or more of these risks or uncertainties materialize, or the underlying estimates or assumptions prove incorrect, actual results may vary significantly from those expressed in such forward-looking statements, and there can be no assurance that the forward-looking statements contained in this report will in fact occur.
| ITEM 1 | BUSINESS |
General
We are a leading developer, manufacturer and distributor of medical equipment for treating, diagnosing, and managing sleep-disordered breathing and other respiratory disorders. Sleep-disordered breathing, or SDB, includes obstructive sleep apnea, or OSA, and other respiratory disorders that occur during sleep. When we were formed in 1989, our primary purpose was to commercialize a treatment for OSA. This treatment, nasal Continuous Positive Airway Pressure, or CPAP, was the first successful noninvasive treatment for OSA. CPAP systems deliver pressurized air, typically through a nasal mask, to prevent collapse of the upper airway during sleep.
Since the development of CPAP, we have developed a number of innovative products for SDB and other respiratory disorders including airflow generators, diagnostic products, mask systems, headgear and other accessories. Our growth has been fuelled by geographic expansion, increased awareness of respiratory conditions as a significant health concern among physicians and patients, and our research and product development efforts.
We employ approximately 3,900 people and sell our products in approximately 100 countries through a combination of wholly owned subsidiaries and independent distributors.
- 2 -
Table of Contents
Our web site address is www.resmed.com. We make our periodic reports, together with any amendments, available on our web site, free of charge, as soon as reasonably practicable after we electronically file or furnish the reports with the Securities and Exchange Commission, or SEC. Information contained on the website is not part of or incorporated into the annual report.
Corporate History
ResMed Inc., a Delaware corporation, was formed in March 1994 as the ultimate holding company for our operating subsidiaries. On June 1, 1995, we completed an initial public offering of common stock and on June 2, 1995 our common stock commenced trading on the NASDAQ National Market. On September 30, 1999 we transferred our principal public listing to the New York Stock Exchange, or NYSE, trading under the ticker symbol RMD. On November 25, 1999, we established a secondary listing of our common stock via Chess Depositary Instruments, or CDIs, on the Australian Stock Exchange (now known as the Australian Securities Exchange), or ASX, also under the symbol RMD. Ten CDIs on the ASX represent one share of our common stock on the NYSE.
Our Australian subsidiary, ResMed Holdings Limited, was originally organized in 1989 by Dr. Peter Farrell to acquire from Baxter Center for Medical Research Pty Limited, or Baxter, the rights to certain technology relating to CPAP treatment as well as Baxters existing CPAP device business. Baxter acquired the rights to the technology in 1987, and sold CPAP devices in Australia from 1988 until our acquisition of the business.
Since formation we have acquired a number of operating businesses including distributors, suppliers and developers of medical equipment.
Segment Information
We believe that, given the single market focus of our operations solely in the sleep-disordered breathing sector of the respiratory medicine industry, and the inter-dependence of its products, we operate as a single operating segment. See Note 15 Segment Information of the Notes to Financial Statements (Part II, Item 8) for financial information regarding segment reporting. Financial information about our revenues from and assets located in foreign countries is also included in the Notes to our consolidated financial statements.
The Market
Sleep is a complex neurological process that includes two distinct states: rapid eye movement, or REM, sleep and non-rapid eye movement, or non-REM, sleep. REM sleep, which is about 20-25% of total sleep experienced by adults, is characterized by a high level of brain activity, bursts of rapid eye movement, increased heart and respiration rates, and paralysis of many muscles. Non-REM sleep is subdivided into four stages that generally parallel sleep depth; stage 1 is the lightest and stage 4 is the deepest.
The upper airway has no rigid support and is held open by active contraction of upper airway muscles. Normally, during REM sleep and deeper levels of non-REM sleep, upper airway muscles relax and the airway narrows. Individuals with narrow upper airways or poor muscle tone are prone to temporary collapses of the upper airway during sleep, called apneas, and to near closures of the upper airway called hypopneas. These breathing events result in a lowering of blood oxygen concentration, causing the central nervous system to react to the lack of oxygen or increased carbon dioxide and signaling the body to respond. Typically, the individual subconsciously arouses from sleep, causing the throat muscles to contract, opening the airway. After a few gasping breaths, blood oxygen levels increase and the individual can resume a deeper sleep until the cycle repeats itself. Sufferers of OSA
- 3 -
Table of Contents
typically experience ten or more such cycles per hour. While these awakenings greatly impair the quality of sleep, the individual is not normally aware of these disruptions. In addition, OSA has recently been recognized as a cause of hypertension and a significant co-morbidity for heart disease, stroke and diabetes.
A 2013 epidemiology study estimated that 26% of adults age 30-70 have some form of obstructive sleep apnea. In the United States alone, this represents approximately 46 million people. Despite the high prevalence of OSA, there is a general lack of awareness of OSA among both the medical community and the general public. It is estimated that less than 20% of those with OSA have been diagnosed or treated. Many healthcare professionals are often unable to diagnose OSA because they are unaware that such non-specific symptoms as excessive daytime sleepiness, snoring, hypertension and irritability are characteristic of OSA.
While OSA has been diagnosed in a broad cross-section of the population, it is predominant among middle-aged men and those who are obese, smoke, consume alcohol in excess or use muscle-relaxing and pain-killing drugs. A strong association has been discovered between OSA and a number of cardiovascular diseases. Studies have shown that SDB is present in approximately 83% of patients with drug-resistant hypertension, approximately 72% of patients with type 2 diabetes, approximately 77% of patients with obesity and approximately 76% of patients with congestive heart failure.
Sleep-Disordered Breathing and Obstructive Sleep Apnea
Sleep-disordered breathing encompasses all disease processes that cause abnormal breathing patterns during sleep. Manifestations include OSA, central sleep apnea, or CSA, and hypoventilation syndromes that occur during sleep. Hypoventilation syndromes are generally associated with obesity, chronic obstructive lung disease and neuromuscular disease. OSA is the most common form of SDB.
Sleep fragmentation and the loss of the deeper levels of sleep caused by OSA can lead to excessive daytime sleepiness, reduced cognitive function, including memory loss and lack of concentration, depression and irritability. OSA sufferers also experience an increase in heart rate and an elevation of blood pressure during the cycle of apneas. Several studies indicate that the oxygen desaturation, increased heart rate and elevated blood pressure caused by OSA may be associated with increased risk of cardiovascular morbidity and mortality due to angina, stroke and heart attack. Patients with OSA have been shown to have impaired daytime performance in a variety of cognitive functions including problem solving, response speed and visual motor coordination, and studies have linked OSA to increased occurrences of traffic and workplace accidents.
Generally, an individual seeking treatment for the symptoms of OSA is referred by a general practitioner to a sleep specialist for further evaluation. The diagnosis of OSA typically requires monitoring the patient during sleep at either a sleep clinic or the patients home. During overnight testing, respiratory parameters and sleep patterns may be monitored, along with other vital signs such as heart rate and blood oxygen levels. Simpler tests, using devices such as our Apnealink, or our automatic positive airway pressure devices, monitor airflow during sleep, and use computer programs to analyze airflow patterns. These tests allow sleep clinicians to detect any sleep disturbances such as apneas, hypopneas or subconscious awakenings.
Existing Therapies
Before 1981, the primary treatment for OSA was a tracheotomy, a surgical procedure to create a hole in the patients windpipe. Alternative surgical treatments have involved either uvulopalatopharyngoplasty, or UPPP, in which surgery is performed on the upper airway to remove excess tissue and to streamline the shape of the airway or implanting a device to add support to the
- 4 -
Table of Contents
soft palate. UPPP alone has a poor success rate; however, when performed in conjunction with multi-stage upper airway surgical procedures, a greater success rate has been claimed. These combined procedures, performed by highly specialized surgeons, are expensive and involve prolonged and often painful recovery periods. Surgical treatments are not considered first line therapy for OSA. Other alternative treatments available today include nasal surgery, mandibular advancement surgery, dental appliances, palatal implants, somnoplasty and nasal devices. Alternative treatments reported to be under development include pharmaceutical therapies and electrical stimulation of the nerves or muscles.
A variety of devices are marketed for the treatment of OSA. Most are only partially effective, but CPAP is a reliable treatment for all severities of OSA and is considered first-line therapy. Use of mandibular advancement devices is increasing as a second-line option in patients unable to use CPAP or those with mild OSA. These devices cause the mandible and tongue to be pulled forward and improve the dimensions of the upper airway. CPAP is a non-invasive means of treating OSA. CPAP was first used as a treatment for OSA in 1980 by Dr. Colin Sullivan, the past Chairman of our Medical Advisory Board and was commercialized for treatment of OSA in the United States in the mid 1980s. During CPAP treatment, a patient sleeps with a nasal interface connected to a small portable airflow generator that delivers room air at a positive pressure. The patient breathes in air from the flow generator and breathes out through an exhaust port in the interface. Continuous air pressure applied in this manner acts as a pneumatic splint to keep the upper airway open and unobstructed. Interfaces include nasal masks and nasal pillows. Sometimes, when a patient leaks air through their mouth, a full-face mask may need to be used, rather than a nasal interface.
CPAP is not a cure and therefore, must be used on a nightly basis as long as treatment is required. Patient compliance has been a major factor in the efficacy of CPAP treatment. Early generations of CPAP units provided limited patient comfort and convenience. Patients experienced soreness from the repeated use of nasal masks and had difficulty falling asleep with the CPAP device operating at the prescribed pressure. In more recent years, product innovations to improve patient comfort and compliance have been developed. These include more comfortable patient interface systems; delay timers that gradually raise air pressure allowing the patient to fall asleep more easily; bilevel air flow generators, including Variable Positive Airway Pressure, or VPAP systems, which provide different air pressures for inhalation and exhalation; heated humidification systems to make the airflow more comfortable; and autotitration devices that reduce the average pressure delivered during the night.
Business Strategy
We believe that the SDB market will continue to grow in the future due to a number of factors including increasing awareness of OSA, improved understanding of the role of SDB treatment in the management of cardiac, neurologic, metabolic and related disorders, and an increase in home-based diagnosis. Our strategy for expanding our business operations and capitalizing on the growth of the SDB market consists of the following key elements:
Continue Product Development and Innovation. We are committed to ongoing innovation in developing products for the diagnosis and treatment of SDB. We have been a leading innovator of products designed to treat SDB more effectively, increase patient comfort and encourage compliance with prescribed therapy. For example, in 2011, we introduced the S9 bilevel range of flow generators, the Quattro FX full face mask, the Swift FX for Her nasal pillow mask, the Mirage FX nasal mask, the Mirage FX for Her nasal mask and the Stellar ventilation device. In 2012, we introduced Swift FX Bella mask, Pixi pediatric mask, Quattro FX for Her and the EasyCare compliance management solution. In 2013, we introduced new products across both our mask and flow generator categories, including the VPAP COPD, Quattro Air, Swift FX Bella, Swift FX Nano and ResMeds SleepSeeker. We believe that continued product development and innovation are key factors to our ongoing
- 5 -
Table of Contents
success. Approximately 15% of our employees are devoted to research and development activities. In fiscal year 2013, we invested $120.1 million, or approximately 8% of our net revenues, in research and development.
Expand Geographic Presence. We market our products in approximately 100 countries to sleep clinics, home healthcare dealers and third-party payers. We intend to increase our sales and marketing efforts in our principal markets, as well as expand the depth of our presence in other geographic regions.
Increase Public and Clinical Awareness. We intend to continue to expand our existing promotional activities to increase awareness of SDB and our treatment alternatives. These promotional activities target both the population with predisposition to SDB and medical specialists, such as cardiologists, neurologists and pulmonologists. In addition, we also target special interest groups, including the National Stroke Association, the American Heart Association and the National Sleep Foundation. In concert with other industry participants, we sponsor educational programs targeted at the primary care physician community, which should further enlighten both doctors and patients about the relationship between SDB or OSA and co-morbidities such as cardiac disease, diabetes, hypertension and obesity. The programs should also support our efforts to inform the community of the dangers of sleep apnea with regard to occupational health and safety, especially in the transport industry.
Expand into New Clinical Applications. We continually seek to identify new applications of our technology for significant unmet medical needs. Studies have established a clinical association between OSA and both stroke and congestive heart failure, and have recognized SDB as a cause of hypertension or high blood pressure. Research also indicates that SDB is independently associated with glucose intolerance and insulin resistance. We have developed a device for the treatment of Cheyne-Stokes breathing in patients with congestive heart failure. In addition, we maintain close working relationships with a number of prominent physicians to explore new medical applications for our products and technology. In 2007, we received Food and Drug Administration, or FDA, clearance and launched a new product in the United States for the treatment of respiratory insufficiency due to central sleep apnea, mixed apnea and periodic breathing, called the Adapt SV. The Adapt SV uses a technology known as adaptive servo-ventilation which utilizes an advanced algorithm to calculate a patient-specific minute ventilation target and automatically adjusts pressure support to maintain the target. We believe this technology has allowed physicians to successfully treat complex breathing disorders in some patients who had previously tried and failed traditional positive airway pressure therapy.
Leverage the Experience of our Management Team. Our senior management team has extensive experience in the medical device industry in general, and in the field of SDB in particular. We intend to continue to leverage the experience and expertise of these individuals to maintain our innovative approach to the development of products and increase awareness of the serious medical problems caused by SDB.
Products
Our portfolio of products includes airflow generators, diagnostic products, mask systems, headgear and other accessories.
Air Flow Generators
We produce CPAP, VPAP and AutoSet systems for the titration and treatment of SDB. The flow generator systems deliver positive airway pressure through a patient interface, either a small nasal mask, nasal pillows system, or full-face mask. Our VPAP units deliver ultra-quiet, comfortable
- 6 -
Table of Contents
bilevel therapy. There are two preset pressures: a higher pressure as the patient breathes in, and a lower pressure as the patient breathes out. Breathing out against a lower pressure makes treatment more comfortable, particularly for patients who need high pressure levels or for those with impaired breathing ability. AutoSet systems are based on a proprietary technology to monitor breathing and can also be used in the diagnosis, treatment and management of OSA.
With the acquisition of ResMed Paris SAS, previously Saime SA, in May 2005, we increased our presence in the European homecare ventilation market. The VS and Elisée range of products are sophisticated, yet easy to use for physicians, clinicians and patients. We believe these devices complement our VPAP Adapt SV and Autoset CS2 for patients who need ventilatory assistance. During fiscal year 2011, we also launched the Stellar 100 and 150 ventilation devices, which provide both invasive and non-invasive ventilation applications for adult and pediatric patients.
Flow generators in total accounted for approximately 54%, 54% and 56% of our net revenues in fiscal years 2013, 2012 and 2011, respectively.
The tables below provide a selection of products, as known by our trademarks, which have been released during the last five years.
|
CONTINUOUS POSITIVE AIRWAY PRESSURE PRODUCTS |
DESCRIPTION | DATE OF COMMERCIAL INTRODUCTION |
||
| S8 Escape (Lightweight) II (ROW, ex Japan) |
A small CPAP device with enhanced feature set to the original S8 Escape (Lightweight), with further improved patient therapy comfort. The device has an optional integrated humidifier. | September 2008 | ||
| S9 Elite |
Premium level CPAP device in ResMeds sleek, compact S9 Series. Features Enhanced Easy-Breathe motor, Expiratory Pressure Relief (EPR) and detailed data options. The device also has an optional integrated humidifier (H5i), ClimateLine heated tube and the small, lightweight SlimLine tube. | February 2010 | ||
| S9 Escape |
As the Standard CPAP model of the S9 Series, the S9 Escape features Expiratory Pressure Relief (EPR) and other innovative features including Climate Control and the enhanced Easy-Breathe motor. The device also has an optional integrated humidifier (H5i). | September 2010 | ||
|
VARIABLE POSITIVE AIRWAY |
DESCRIPTION | DATE OF COMMERCIAL INTRODUCTION |
||
|
VPAP III STA with QuickNav |
An upgraded Bi-level device with alarm history, instant efficacy data and a large screen. | July 2008 | ||
| VPAP S / VPAP IV |
Bi-level device that provides S and CPAP modes with the pressure up to 25 cmH2O in a compact and convenient S8 design. | September 2008 | ||
- 7 -
Table of Contents
|
VARIABLE POSITIVE AIRWAY |
DESCRIPTION | DATE OF COMMERCIAL INTRODUCTION |
||
| VPAP IV ST# |
Small compact Bi-level ST device in an S8 box with VAuto for Europe. | September 2008 | ||
| S8 Auto 25 |
Bi-level device that provides the Easy-Breathe wave on the AutoSet algorithm and the pressure up to 25cm H2O in a compact and convenient S8 design. | October 2008 | ||
| VPAP Tx Lab System | VPAP Tx therapy device features all ResMeds sleep therapy modes. Tx Link connection module relays signals from the device to PSG equipment. The system is controlled through the user-friendly EasyCare Tx titration software. | March 2010 | ||
| S9 VPAP S |
Bilevel pressure support therapy device in ResMeds sleek, compact S9 Series. Designed for comfort and compliance with the Easy-Breath waveform in S-mode* and pressures up to 25 cmH2O. The device also has an optional integrated humidifier (H5i), ClimateLine heated tube and the small, lightweight SlimLine tube. *Americas only | March 2011 | ||
| S9 VPAP ST |
Bilevel pressure support therapy device with pressures up to 25 cmH2O designed for comfort, effective therapy with the assurance of back up rate up to 50 bpm. The device also has an optional integrated humidifier (H5i), ClimateLine heated tube and the small, lightweight SlimLine tube. | March 2011 | ||
| S9 VPAP Auto |
Premium auto-adjusting device with the unique VAuto mode and Easy-Breathe technology designed for patients requiring both higher pressures and pressure relief. VAuto mode features enhanced AutoSet technology with central sleep apnea (CSA) detection. The device may be used with an optional integrated humidifier (H5i), ClimateLine heated tube and the small, lightweight SlimLine tube | March 2011 | ||
| S9 VPAP Adapt |
Adaptive Servo-Ventilator specifically designed to provide a rapid response to periodic breathing for the treatment of central and/or mixed apneas, providing ventilatory support when it is needed packaged in ResMeds sleek, compact S9 Series. The device also offers an optional integrated humidifier (H5i), ClimateLine heated tube and the small, lightweight SlimLine tube. | March 2011 | ||
- 8 -
Table of Contents
|
VARIABLE POSITIVE AIRWAY PRESSURE PRODUCTS |
DESCRIPTION | DATE OF COMMERCIAL INTRODUCTION |
||
| S9 AutoSet CS# |
Adaptive Servo-Ventilator specifically designed to provide a rapid response to Cheyne-Stokes breathing and periodic breathing associated with Heart Failure for the treatment of central and/or mixed apneas, providing ventilatory support when it is needed. Packaged in ResMeds sleek, compact S9 Series. The device also has an optional integrated humidifier (H5i), ClimateLine heated tube and the small, lightweight SlimLine tube. | March 2011 | ||
| S9 Auto 25# |
Premium auto-adjusting device with the unique VAuto mode and Easy-Breathe technology designed for patients requiring both higher pressures and pressure relief. VAuto mode features enhanced AutoSet technology with central sleep apnea (CSA) detection. The device may be used with an optional integrated humidifier (H5i), ClimateLine heated tube and the small, lightweight SlimLine tube | March 2011 | ||
| S9 VPAP COPD |
Bilevel pressure support up to pressure 30cmH2O with both fixed and adjustable alarms. This device has been specifically designed for COPD. | April 2013 | ||
# Sold outside United States only
|
AUTOMATIC POSITIVE AIRWAY PRESSURE PRODUCTS |
DESCRIPTION | DATE OF COMMERCIAL INTRODUCTION |
||
| S9 AutoSet |
Premium APAP device packaged in ResMeds sleek, compact S9 Series. Features Enhanced AutoSet (with Central Sleep Apnea Detection), Enhanced Easy-Breathe motor, expiratory pressure relief (EPR) and detailed data options. The device also has, an optional integrated humidifier (H5i), ClimateLine heated tube and the small, lightweight SlimLine tube. | February 2010 | ||
| S9 Escape Auto |
The S9 Escape Auto is the Standard APAP device packaged in ResMeds sleek, compact S9 Series. It features an intelligent algorithm with Easy-Breathe expiratory pressure relief (EPR) and delivers whisper-quiet therapy in a smooth waveform. The device also offers an optional integrated humidifier (H5i), Climate Control with the ClimateLine heated tube and the small, lightweight SlimLine tube. | September 2010 | ||
- 9 -
Table of Contents
| VENTILATION PRODUCTS | DESCRIPTION | DATE OF COMMERCIAL INTRODUCTION |
||
| Elisée 150*# |
New software launch V2.50 incorporating CPAP mode and additional flexibility in settings. For example presetting 2 programs in both invasive and non-invasive. | November 2008 | ||
| VS III *# |
Pressure support and volume ventilator for invasive and non-invasive purposes so it can be used from the hospital to the home. Launched in France and Germany. | December 2008 | ||
|
Stellar 100 and 150 # |
Pressure support and volume ventilator for invasive and non-invasive purposes so it can be used from the hospital to the home. | March 2011 | ||
* Not cleared for marketing in the United States
# Sold outside United States only
Masks, Accessories, Motors and Diagnostic Products
Masks, accessories, motors and diagnostic products together accounted for approximately 46%, 46% and 44% of our net revenues in fiscal years 2013, 2012 and 2011, respectively.
Mask Systems and Diagnostic Products
Mask systems are one of the most important elements of SDB treatment systems. Masks are a primary determinant of patient comfort and as such may drive or impede patient compliance with therapy. We have been a consistent innovator in masks, improving patient comfort while minimizing size and weight.
| MASK PRODUCTS | DESCRIPTION | DATE OF COMMERCIAL INTRODUCTION |
||
| Activa LT | Nasal mask including Active Cell Technology in a lightweight version to help mitigate leak and optimize patient comfort | October 2008 | ||
| Swift LT for Her | Nasal mask offering pillows systems with female specific design features | November 2008 | ||
| Swift FX | Fourth generation nasal pillows system offering a fully flexible design for comfort and performance | September 2009 | ||
| Mirage SoftGel | Nasal mask offering a gel cushion, interchangeable with the Activa LT system to improve choice and comfort | October 2009 | ||
| Quattro FX | Full Face mask offering unobtrusive fit | September 2010 | ||
| Swift FX for Her | Fourth generation nasal pillows system offering a fully flexible design for comfort and performance with female specific design features | September 2010 | ||
- 10 -
Table of Contents
| MASK PRODUCTS | DESCRIPTION | DATE OF COMMERCIAL INTRODUCTION |
||
| Mirage FX | Nasal mask offering auto adjusting forehead support and SoftEdge headgear | October 2010 | ||
| Mirage FX for Her | Nasal mask offering auto adjusting forehead support and SoftEdge headgear with female specific design features | April 2011 | ||
| Pixi Pediatric Mask | A pediatric mask designed for children 2 years and older | September 2011 | ||
| Quattro FX for Her | Full face mask offering unobtrusive fit with female specific design features | October 2011 | ||
| Swift FX Bella | Fourth generation nasal pillows system with an alternative headgear design | January 2012 | ||
| Quattro Air | Next Generation lightweight Full Face Mask with improved comfort | June 2013 | ||
| Swift FX Nano | A compact nasal mask designed to deliver an excellent user experience, without compromising on fit, comfort and ease of use. | June 2013 | ||
We market sleep recorders for the diagnosis and titration of SDB in sleep clinics and hospitals. These diagnostic systems record relevant respiratory and sleep data, which can be analyzed by a sleep specialist or physician who can then tailor an appropriate OSA treatment regimen for the patient.
| DIAGNOSTIC PRODUCTS | DESCRIPTION | DATE OF COMMERCIAL INTRODUCTION |
||
| ApneaLink + Oximetry | A portable diagnostic device with oximetry measurement | June 2007 | ||
| ApneaLink Plus (U.S.) | A portable diagnostic device with oximetry measurement and respiratory effort measurement | June 2009 | ||
- 11 -
Table of Contents
Accessories and Other Products
To assist those professionals diagnosing or managing the treatment of patients there are data communications and control products such as EasyCare, ResLink, ResControl, ResControl II, TxControl, ResScan and ResTraxx modules that facilitate the transfer of data and other information to and from the flow generators. To enhance patient comfort, convenience and compliance, we market a variety of other products and accessories. These products include humidifiers, such as H5i and H4i, which connect directly with the CPAP, VPAP and AutoSet flow generators to humidify and heat the air delivered to the patient, helping to prevent the drying of nasal passages that can cause discomfort. Other optional accessories include cold passover humidifiers, carry bags and breathing circuits.
|
DATA / PATIENT MANAGEMENT PRODUCTS |
DESCRIPTION | DATE OF COMMERCIAL INTRODUCTION |
||
| S9 Embletta Adapter |
The S9 Embletta Adapter provides a connection between an S9 device and an Embletta Portable Diagnostic System | November 2010 | ||
| ResScan v3.14 |
An easy and flexible patient monitoring system providing therapy insights. This version included support for S9 bilevel and cross-patient first 30 days compliance reporting. | April 2011 | ||
| ResTraxx v17.1 |
ResMeds web-based compliance monitoring system which introduced several new features to ResTraxx Online reports and enhanced support for S9 VPAP devices. | April 2011 | ||
| ResTraxx v 18.3 |
ResMeds web-based compliance monitoring system introducing EasyCare Card online compliance reporting direct from device SD card to ResTraxx Online | November 2011 | ||
| ResScan V3.16 |
ResMeds easy and flexible patient monitoring system providing therapy insights and supporting VS and Elise ventilation products (Europe) | November 2011 | ||
| EasyCare 1.0 |
ResMeds new compliance management solution offers both wireless and card-to-cloud functionality, providing access to patient data anywhere with an internet connection. Intuitive user interface, easy to understand reports and automated compliance notification. | April 2012 | ||
Product Development and Clinical Trials
We have a strong track record in innovation in the sleep market. In 1989, we introduced our first CPAP device. Since then we have been committed to an ongoing program of product advancement and development. Currently, our product development efforts are focused on not only improving our current product offerings, but also expanding into new product applications.
We continually seek to identify new applications of our technology for significant unmet medical needs. SDB is associated with a number of symptoms beyond excessive daytime sleepiness and irritability. Studies have established a clinical association between SDB and hypertension, stroke,
- 12 -
Table of Contents
congestive heart failure and diabetes. We support clinical trials in many countries including the United States, Germany, France, the United Kingdom, Italy, Switzerland, China and Australia to develop new clinical applications for our technology.
We consult with physicians at major sleep centers throughout the world to identify technological trends in the treatment of SDB. New product ideas are also identified by our marketing staff, direct sales force, network of distributors, customers and patients.
In fiscal years 2013, 2012 and 2011 we invested $120.1 million, $109.7 million and $92.0 million, respectively, on research and development.
Sales and Marketing
We currently market our products in approximately 100 countries through a network of distributors and our direct sales force. We attempt to tailor our marketing approach to each national market, based on regional awareness of SDB as a health problem, physician referral patterns, consumer preferences and local reimbursement policies. See Note 15 Segment Information of the Notes to Consolidated Financial Statements (Part II, Item 8) for financial information about our geographic areas.
North America and Latin America. Our products are typically purchased by a home healthcare dealer who then sells the products to the patient. The decision to purchase our products, as opposed to those of our competitors, is made or influenced by one or more of the following individuals or organizations: the prescribing physician and his or her staff; the home healthcare dealer; the insurer and the patient. In North and Latin America, our sales and marketing activities are conducted through a field sales organization made up of regional territory representatives, program development specialists and regional sales directors. Our field sales organization markets and sells products to home healthcare dealer branch locations throughout the North and Latin America.
We also market our products directly to sleep clinics. Patients who are diagnosed with OSA and prescribed CPAP treatment are typically referred by the diagnosing sleep clinic to a home healthcare dealer to fill the prescription. The home healthcare dealer, in consultation with the referring physician, will assist the patient in selecting the equipment, fit the patient with the appropriate mask and set the flow generator pressure to the prescribed level.
Sales in North and Latin America accounted for 56%, 55% and 53% of our net revenues for fiscal years 2013, 2012 and 2011, respectively.
Europe. We market our products in most major European countries. We have wholly-owned subsidiaries in Austria, Finland, France, Germany, Norway, Netherlands, Sweden, Switzerland, the United Kingdom and Ireland. We use independent distributors to sell our products in other areas of Europe. Distributors are selected in each country based on their knowledge of respiratory medicine and a commitment to SDB therapy. In each country in which we sell our products direct, a local senior manager is responsible for direct national sales. In many countries in Europe, we sell our products to home healthcare dealers who then sell the products to the patients. In Germany, we also operate a home healthcare company, in which we provide products and services directly to patients, and receive reimbursement directly from third-party payers.
Sales in Europe accounted for 33%, 35% and 37% of our total net revenues for fiscal years 2013, 2012 and 2011, respectively.
Asia Pacific. We have wholly-owned subsidiaries in Australia, Hong Kong, Japan, New Zealand, China and India. We use a combination of our direct sales force and independent distributors to sell our products in Asia Pacific. Sales in Asia Pacific accounted for 11%, 10% and 10% of our total net revenues for the fiscal years 2013, 2012 and 2011, respectively.
- 13 -
Table of Contents
Other Marketing Efforts. We continue to pursue suitable opportunities with professional and healthcare associations to raise awareness of the importance of SDB in cardiology patients, including coronary artery disease, congestive heart failure, hypertension and stroke. Clinical research over the past decade has demonstrated a high prevalence of OSA in cardiology patients and has suggested that it may increase the risk of developing cardiovascular disease and heart failure. In September 2008, the European Society of Cardiologists published guidelines for the treatment of acute and chronic heart failure. The guidelines noted that patients with symptomatic heart failure frequently have sleep-related disorders (central or obstructive sleep apnea) and recommended treatment with Continuous Positive Airway Pressure, or CPAP, for patients diagnosed with obstructive sleep apnea. In 2013, the American College of Cardiology Foundation and the American Heart Association issued guidelines for the management of heart failure. Those guidelines recognized that CPAP can be beneficial to improve functional status in patients with heart failure and sleep apnea. We are conducting several clinical studies investigating the role of OSA in cardiology diseases and are engaged with professional bodies to increase awareness of OSA amongst cardiologists.
We also continue to work to raise awareness of SDB in diabetes. Current research is increasingly showing an independent association between OSA and type 2 diabetes and there is preliminary evidence that OSA may worsen diabetes control. Accordingly, we initiated a study investigating the prevalence of OSA in the type 2 diabetic population. Due to the high prevalence of the SDB and type 2 diabetes, we are now actively supporting the American Association of Diabetes Educators and are in the process of setting up further initiatives to develop the SDB market in the diabetic population. ResMed is also reaching out to diabetes patients through our online partners. ResMed is educating people who suffer from diabetes about the overlap with obstructive sleep apnea and directing them via www.Healthysleep.com to ResMed partner sleep centers.
In June 2008, the International Diabetes Federation, or IDF, released a statement on SDB and type 2 diabetes. The IDF Taskforce on Epidemiology and Prevention strongly recommended that health professionals working in both type 2 diabetes and SDB adopt clinical practices to ensure that a patient presenting with one condition is considered for the other. Furthermore, the IDF recommended that people with type 2 diabetes should be screened for OSA particularly when they present classical symptoms such as witnessed apneas, heavy snoring or daytime sleepiness and poor workplace performance. In March 2011, the American Association of Clinical Endocrinologists published updated medical guidelines for developing a comprehensive care plan for patients with diabetes, recommending screening for OSA/SDB in adults with type 2 diabetes, especially men older than 50 years.
In April 2010, the National Institutes of Health released a clinical study reporting that obstructive sleep apnea is associated with an increased risk of stroke in middle-aged and older adults, especially men. In a recently released study in Circulation, it was reported that obstructive sleep apnea is associated with an increased risk of incident heart failure in a general community of middle-aged and older men. Specifically, men ages 40 to 70 with an apnea-hypopnea index, or AHI, of equal to or greater than 30 were 68% more likely to develop coronary heart disease than those with AHI of less than 5.
We are working with the anesthesiology community to help reduce the potential for peri-operative deaths due to undiagnosed sleep-disordered breathing in patients undergoing surgical procedures. We continue to provide research funding in these strategic areas while at the same time providing educational support to physicians working within these various specialties
We believe that the increasing awareness among physicians supports the efforts and investment we are making in new markets, including diabetes and cardiology.
- 14 -
Table of Contents
Manufacturing
Our manufacturing operations consist primarily of assembly and testing of our flow generators, masks and accessories. Of the numerous raw materials, parts and components purchased for assembly of our therapeutic and diagnostic sleep disorder products, most are off-the-shelf items available from multiple vendors. We generally manufacture to our internal sales forecasts and fill orders as received. Over the last few years, the manufacturing processes have been transformed along lean manufacturing guidelines to flow lines staffed by dedicated teams. Each team is responsible for the manufacture and quality of their product group and decisions are based on performance and quality measures, including customer feedback.
Our principal manufacturing facility is located in Sydney, Australia and comprises a 155,000 square foot manufacturing facility. We have a 174,000 square foot assembly and distribution facility in South Carolina; the plant specializes in regional customization of our flow generators. We have a 95,000 square foot manufacturing facility in Singapore to complement the Sydney manufacturing site. The plant assembles masks, flow generators and electric motors. We have a 46,000 square foot manufacturing facility in Malaysia. The plant specializes in the manufacture of headgear material for our masks and accessories. We have a 43,000 square foot manufacturing facility in Paris, France. The facility is primarily responsible for the assembly of mechanical ventilators and associated accessories. We have a 43,000 square foot manufacturing facility in Freudenstadt, Germany; the plant specializes in the manufacture of medical humidification products. We also manufacture high-quality electric motors for our flow generator devices at a 72,000 square foot manufacturing facility in Chatsworth, California.
Our quality management system is based upon the requirements of ISO 9001, ISO 13485, FDA Quality System Regulations for Medical Devices, the Medical Device Directive (93/42/EEC) and other applicable regulations for the markets in which we sell. All of our manufacturing sites are accredited to ISO 13485. These sites are subject to third-party audits, conducted by the ISO notified bodies, at regular intervals.
Third-Party Coverage and Reimbursement
The cost of medical care in many of the countries in which we operate is funded in substantial part by government and private insurance programs. In Germany, we receive payments directly from these payers. Outside Germany, although we do not generally receive payments for our products directly from these payers, our success in major markets is dependent upon the ability of patients to obtain coverage and adequate reimbursement from third-party payers for our products.
In the United States, our products are purchased primarily by home healthcare dealers, hospitals or sleep clinics, which then invoice third-party payers directly for reimbursement. Domestic third-party payers include Medicare, Medicaid and corporate health insurance plans. These payers may deny coverage and reimbursement if they determine that a device is not used in accordance with certain covered treatment methods, or is experimental, unnecessary or inappropriate. The long-term trend towards cost-containment, through managed healthcare, or other legislative proposals to reform healthcare, could control or significantly influence the purchase of healthcare services and products and could result in lower prices for our products. In some foreign markets, such as France, Germany and Japan, government reimbursement is currently available for purchase or rental of our products, subject to constraints such as price controls or unit sales limitations. In Australia and in some other foreign markets, there is currently limited or no reimbursement for devices that treat OSA.
The past decade of legislative reform in the United States, including the 2010 Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act
- 15 -
Table of Contents
(collectively, the PPACA), Medicare Improvement for Patients and Providers Act of 2008 (MIPPA), Deficit Reduction Act of 2005 (DRA), and the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (MMA), has significantly impacted reimbursement for products that we provide. The longer term impact, though not entirely predictable, continues to bring significant changes to the third-party payer landscape.
Beginning in 2005, the MMA reduced payment amounts for five categories of HME, froze payment amounts for certain covered home medical equipment (HME) items through 2007, established a Medicare competitive acquisition program for HME and implemented quality standards and accreditation requirements for HME suppliers. The DRA capped the Medicare rental period for certain capped rental items, including CPAP devices, at 13 months of continuous use, after which time title of the equipment would transfer automatically to the beneficiary. MIPPA retroactively delayed the implementation of competitive bidding for eighteen months and decreased the 2009 Medicare fee schedule payment amounts for HME by 9.5% for product categories included in competitive bidding. Because the annual update factor for 2010 was 0.0 %, the 2009 fee schedule payment rates remained effective for 2010. For 2011, the fee schedule amounts were reduced by 0.1%, and for 2012, the fee schedule amounts were increased by 2.4% and for 2013, the fee schedule amounts were increased by 0.8%.
Effective January 1, 2011, the Centers for Medicare & Medicaid Services, or CMS, implemented the first round of competitive bidding program in 9 competitive bidding areas, or CBAs and included home medical equipment such as oxygen and oxygen equipment, CPAP and respiratory assist devices, and related supplies and accessories. The average reduction from current Medicare payment rates in this first round of competitive bidding was approximately 32% overall and 34% for CPAP and respiratory devices. On January 30, 2013, CMS announced the single payment amounts for the second round, which cover a total of 91 CBAs. For CPAP and respiratory devices, the average reduction from current Medicare payment rates in the second round was approximately 47% on a weighted average basis, effective July 1, 2013.
The PPACA, which was passed both to expand the number of individuals with healthcare coverage and to develop additional revenue sources, includes, among other things, a deductible excise tax equal to 2.3% of the price for which medical devices are sold in the United States on any entity that manufactures or imports medical devices, with limited exceptions, beginning in 2013. This excise tax is applicable to our products that are primarily used in hospitals and sleep labs, which includes the ApneaLink, VPAP Tx, certain Respiratory Care and dental sleep products. The PPACA also provides for a number of Medicare regulatory requirements, including new face-to-face encounter requirements for durable medical equipment and home health services; and a requirement that by 2016, the competitive bidding process must be rolled-out nationally or prices in non-competitive bidding areas must be adjusted to match competitive bidding prices.
We cannot predict the impact that any U.S. legislation enacted in the future will have on our revenues, profit margins, profitability, operating cash flows and results of operations.
Even though we do not submit claims or bill governmental programs and other third-party payers directly for reimbursement for our products sold in the United States, we are still subject to a number of laws and regulations relating to governmental programs, and any violation of these laws and regulations could result in civil and criminal penalties, including fines. In particular, the federal Anti-Kickback Law prohibits persons from knowingly and willfully soliciting, receiving, offering or providing remuneration, directly or indirectly, to induce either the referral of an individual, or the furnishing, recommending or arranging for a good or service, for which payment may be made under a Federal healthcare program such as the Medicare and Medicaid programs. The government has interpreted this law broadly to apply to the marketing and sales activities of manufacturers and
- 16 -
Table of Contents
distributors like us. Many states have adopted laws similar to the federal Anti-Kickback Law. We are also subject to other federal and state fraud laws applicable to payment from any third-party payer. These laws prohibit persons from knowingly and willfully filing false claims or executing a scheme to defraud any healthcare benefit program, including private third-party payers. These laws may apply to manufacturers and distributors who provide information on coverage, coding and reimbursement of their products to persons who bill third-party payers. We continuously strive to comply with these laws and believe that our arrangements do not violate these laws. Liability may still arise from the intentions or actions of the parties with whom we do business or from a different governmental agency interpretation of the laws.
Service and Warranty
We generally offer either one-year or two-year limited warranties on our flow generator products. Warranties on mask systems are for 90 days. Our distributors either repair our products with parts supplied by us or arrange shipment of products to our facilities for repair or replacement.
We receive returns of our products from the field for various reasons. We believe that the level of returns experienced to date is consistent with levels typically experienced by manufacturers of similar devices. We provide for warranties and returns based on historical data.
Competition
The markets for our products are highly competitive. We believe that the principal competitive factors in all of our markets are product features, reliability and price. Customer support, reputation and efficient distribution are also important factors.
We compete on a market-by-market basis with various companies, some of which have greater financial, research, manufacturing and marketing resources than us. In the United States, our principal market, the primary competitors for our products are: Philips BV; DeVilbiss Healthcare,; and Fisher & Paykel Healthcare Corporation Limited. Our principal international competitors are also Philips BV, DeVilbiss, and Fisher & Paykel Healthcare, plus Apex Medical Corporation, BMC Medical Co. Ltd., as well as regional manufacturers. The disparity between our resources and those of our competitors may increase as a result of the trend towards consolidation in the healthcare industry. In addition, some of our competitors, such as Weinmann Geräte für Medizin GmbH + Co. KG, are affiliates of customers of ours, which may make it difficult to compete with them. Finally, our products compete with surgical procedures and dental appliances designed to treat OSA and other SDB-related respiratory conditions. The development of new or innovative procedures or devices by others could result in our products becoming obsolete or noncompetitive, which would harm our revenues and financial condition.
Any product developed by us that gains regulatory clearance will have to compete for market acceptance and market share. An important factor in such competition may be the timing of market introduction of competitive products. Accordingly, the speed with which we can develop products, complete clinical testing and regulatory clearance processes and supply commercial quantities of the product to the market are important competitive factors. In addition, our ability to compete will continue to be dependent on successfully protecting our patents and other intellectual property.
Patents and Proprietary Rights and Related Litigation
We rely on a combination of patents, trade secrets, copyrights, trademarks and non-disclosure agreements to protect our proprietary technology and rights.
- 17 -
Table of Contents
Through our subsidiaries ResMed Limited, ResMed R&D Germany GmbH, ResMed Motor Technologies Inc., ResMed Sensor Technologies Limited, ResMed SAS, and ResMed Paris SAS, as of the date of this annual report, we own or have licensed rights to approximately 743 issued United States patents (including approximately 342 design patents) and approximately 1,199 issued foreign patents. In addition, there are approximately 449 pending United States patent applications (including approximately 54 design patent applications), approximately 844 pending foreign patent applications, approximately 1,172 registered foreign designs and 1 pending foreign design. Some of these patents, patent applications and designs relate to significant aspects and features of our products.
Of our patents, 124 United States patents and 185 foreign patents are due to expire in the next five years. There are 19 foreign patents due to expire in 2014, 36 in 2015, 5 in 2016, 28 in 2017, and 97 in 2018. There are 7 United States patents due to expire in 2014, 21 United States patents in 2015, 7 United States patents in 2016, 28 United States patents in 2017, and 61 United States patents in 2018. We believe that the expiration of these patents will not have a material adverse impact on our competitive position.
Litigation may be necessary to enforce patents issued to us, to protect our rights, or to defend third-party claims of infringement by us of the proprietary rights of others. The defense and prosecution of patent claims, including pending claims, as well as participation in other inter-party proceedings, can be expensive and time-consuming, even in those instances in which the outcome is favorable to us. Patent laws regarding the enforceability of patents vary from country to country. Therefore, there can be no assurance that patent issues will be uniformly resolved, or that local laws will provide us with consistent rights and benefits.
Government Regulations
Our products are subject to extensive regulation particularly as to safety, efficacy and adherence to FDA Quality System Regulation, and related manufacturing standards. Medical device products are subject to rigorous FDA and other governmental agency regulations in the United States and similar regulations of foreign agencies abroad. The FDA regulates the introduction, manufacture, advertising, labeling, packaging, marketing, distribution and record keeping for such products, in order to ensure that medical products distributed in the United States are safe and effective for their intended use. In addition, the FDA is authorized to establish special controls to provide reasonable assurance of the safety and effectiveness of most devices. Non-compliance with applicable requirements can result in import detentions, fines, civil penalties, injunctions, suspensions or losses of regulatory approvals, recall or seizure of products, operating restrictions, refusal of the government to approve product export applications or allow us to enter into supply contracts, and criminal prosecution.
Unless an exemption applies, the FDA requires that a manufacturer introducing a new medical device or a new indication for use of an existing medical device obtain either a Section 510(k) premarket notification clearance or a premarket approval, or PMA, before introducing it into the U.S. market. Our products currently marketed in the United States are marketed in reliance on 510(k) pre-marketing clearances as either Class I or Class II devices. The process of obtaining a Section 510(k) clearance generally requires the submission of performance data and often clinical data, which in some cases can be extensive, to demonstrate that the device is substantially equivalent to a device that was on the market before 1976 or to a device that has been found by the FDA to be substantially equivalent to such a pre-1976 device. As a result, FDA clearance requirements may extend the development process for a considerable length of time. In addition, in some cases, the FDA may require additional review by an advisory panel, which can further lengthen the process. The PMA process, which is reserved for new devices that are not substantially equivalent to any predicate device and for high-risk devices or those that are used to support or sustain human life, may take several years and requires the submission of extensive performance and clinical information.
- 18 -
Table of Contents
As a medical device manufacturer, all of our domestic and Australian manufacturing facilities are subject to inspection on a routine basis by the FDA. We believe that our design, manufacturing and quality control procedures are in compliance with the FDAs regulatory requirements.
Sales of medical devices outside the United States are subject to regulatory requirements that vary widely from country to country. Approval for sale of our medical devices in Europe is through the CE mark process. Where appropriate, our products are CE marked to the European Unions Medical Device Directive. Under the CE marketing scheme, our products are classified as either Class I or Class II. Our devices are listed in Australia with the Therapeutic Goods Administration, and in Canada with Health Canada.
Employees
As of June 30, 2013, we had approximately 3,900 employees or full-time consultants, of which approximately 1,500 persons were employed in warehousing and manufacturing, 600 in research and development and 1,800 in sales, marketing and administration. Of our employees and consultants, approximately, 1,250 were located in Australia, 800 in North and Latin America, 1,300 in Europe and 550 in Asia.
We believe that the success of our business will depend, in part, on our ability to attract and retain qualified personnel.
| ITEM 1A | RISK FACTORS |
Before deciding to purchase, hold or sell our common stock, you should carefully consider the risks described below in addition to the other cautionary statements and risks described elsewhere, and the other information contained, in this Report and in our other filings with the SEC, including our subsequent reports on Forms 10-Q and 8-K. The risks and uncertainties described below are not the only ones we face. Additional risks and uncertainties not presently known to us or that we currently deem immaterial may also affect our business. If any of these known or unknown risks or uncertainties actually occurs with material adverse effects on us, our business, financial condition and results of operations could be seriously harmed. In that event, the market price for our common stock will likely decline, and you may lose all or part of your investment.
Our inability to compete successfully in our markets may harm our business. The markets for our SDB products are highly competitive and are characterized by frequent product improvements and evolving technology. Our ability to compete successfully depends, in part, on our ability to develop, manufacture and market innovative new products. The development of innovative new products by our competitors or the discovery of alternative treatments or potential cures for the conditions that our products treat could make our products noncompetitive or obsolete. Current competitors, new entrants, academics, and others are trying to develop new devices, alternative treatments or cures, and pharmaceutical solutions to the conditions our products treat.
Additionally, some of our competitors have greater financial, research and development, manufacturing and marketing resources than we do. The past several years have seen a trend towards consolidation in the healthcare industry and in the markets for our products. Industry consolidation could result in greater competition if our competitors combine their resources, if our competitors are acquired by other companies with greater resources than ours, or if our competitors become affiliated with customers of ours. This competition could increase pressure on us to reduce the selling prices of our products or could cause us to increase our spending on research and development and sales and marketing. If we are unable to develop innovative new products, maintain competitive pricing, and offer products that consumers perceive to be as good as those of our competitors, our sales or gross margins could decrease which would harm our business.
- 19 -
Table of Contents
Our business depends on our ability to market effectively to dealers of home healthcare products and sleep clinics. We market our products primarily to home healthcare dealers and to sleep clinics that diagnose OSA and other sleep disorders, as well as to non-sleep specialist physician practices that diagnose and treat sleep disorders. We believe that these groups play a significant role in determining which brand of product a patient will use. The success of our business depends on our ability to market effectively to these groups to ensure that our products are properly marketed and sold by these third-parties.
We have limited resources to market to the sleep clinics, home healthcare dealer branch locations and to the non-sleep specialists, most of whom use, sell or recommend several brands of products. In addition, home healthcare dealers have experienced price pressures as government and third-party reimbursement has declined for home healthcare products, and home healthcare dealers are requiring price discounts and longer periods of time to pay for products purchased from us. We cannot assure you that physicians will continue to prescribe our products, or that home healthcare dealers or patients will not substitute competing products when a prescription specifying our products has been written.
We have expanded our marketing activities to target the population with a predisposition to sleep-disordered breathing as well as primary care physicians and various medical specialists. We cannot assure you that these marketing efforts will be successful in increasing awareness or sales of our products.
If we are unable to support our continued growth, our business could suffer. We have experienced rapid and substantial growth. As we continue to grow, the complexity of our operations increases, placing greater demands on our management. Our ability to manage our growth effectively depends on our ability to implement and improve our financial and management information systems on a timely basis and to effect other changes in our business including, the ability to monitor and improve manufacturing systems, information technology, and quality and regulatory compliance systems, among others. Unexpected difficulties during expansion, the failure to attract and retain qualified employees, the failure to successfully replace or upgrade our management information systems, the failure to manage costs or our inability to respond effectively to growth or plan for future expansion could cause our growth to stop. If we fail to manage our growth effectively and efficiently, our costs could increase faster than our revenues and our business results could suffer.
If we fail to integrate our recent acquisitions with our operations, our business could suffer. We continue to integrate our recent acquisitions into our operations and we may find it difficult to integrate the operations as personnel may leave and licensees, distributors or suppliers may terminate their arrangements or demand amended terms to these arrangements. Additionally, our management may have their attention diverted while trying to integrate these businesses. If we are not able to successfully integrate the operations, we may not realize the anticipated benefits of the acquisitions.
We are subject to various risks relating to international activities that could affect our overall profitability. We manufacture substantially all of our products outside the United States and sell a significant portion of our products in non-U.S. markets. Sales outside North and Latin America accounted for approximately 44% and 46% of our net revenues in the years ended June 30, 2013 and 2012, respectively. We expect that sales within these areas will account for approximately 45% of our net revenues in the foreseeable future. Our sales and operations outside of the U.S. are subject to several difficulties and risks that are separate and distinct from those we face in the U.S., including:
| | fluctuations in currency exchange rates; |
| | tariffs and other trade barriers; |
| | compliance with foreign medical device manufacturing regulations; |
- 20 -
Table of Contents
| | difficulty in enforcing agreements and collecting receivables through foreign legal systems; |
| | reduction in third-party payer reimbursement for our products; |
| | inability to obtain import licenses; |
| | changes in trade policies and in U.S. and foreign tax policies; |
| | possible changes in export or import restrictions; and |
| | the modification or introduction of other governmental policies with potentially adverse effects. |
Any of the above factors may have a material adverse effect on our ability to increase or maintain our non-U.S sales.
Government and private insurance plans may not adequately reimburse our customers for our products, which could result in reductions in sales or selling prices for our products. Our ability to sell our products depends in large part on the extent to which coverage and reimbursement for our products will be available from government health administration authorities, private health insurers and other organizations. These third-party payers are increasingly challenging the prices charged for medical products and services and can, without notice, deny coverage for treatments that may include the use of our products. Therefore, even if a product is approved for marketing, we cannot make assurances that coverage and reimbursement will be available for the product, that the reimbursement amount will be adequate or that the reimbursement amount, even if initially adequate, will not be subsequently reduced. For example, in some markets, such as Spain, France and Germany, government coverage and reimbursement are currently available for the purchase or rental of our products but are subject to constraints such as price controls or unit sales limitations. In other markets, such as Australia, there is currently limited or no reimbursement for devices that treat SDB conditions. As we continue to develop new products, those products will generally not qualify for coverage and reimbursement until they are approved for marketing, if at all.
In the United States, we sell our products primarily to home healthcare dealers, hospitals and to sleep clinics. Reductions in reimbursement to our customers by third-party payers, if they occur, may have a material impact on our customers and, therefore, may indirectly affect our sales to, or the collectability of receivables we have from, those customers. A recent development affecting reimbursement negatively stems from the Medicare Prescription Drug, Improvement, and Modernization Act of 2003, or MMA, which instructed the Centers for Medicare & Medicaid Services, or CMS, the agency responsible for administering the Medicare program, to establish and implement programs under which our customers who provide home healthcare services must compete to offer products in designated competitive bidding areas, or CBAs. CMS recently rolled out the competitive bidding program in 9 CBAs and included home medical equipment such as oxygen and oxygen equipment, CPAP and respiratory assist devices, and related supplies and accessories. On July 2, 2010, CMS announced the single payment amount the amount paid to successful bidders for the first round of the competitive bidding and began offering contracts to qualifying home health companies, effective January 1, 2011. The average reduction from current Medicare payment rates in this first round of competitive bidding was approximately 32% overall and 34% for CPAP and respiratory devices and became effective January 1, 2011. On January 30, 2013, CMS announced the single payment amounts for the second round, which cover a total of 91 CBAs. For CPAP and respiratory devices, the average reduction from current Medicare payment rates in the second round was approximately 47% on a weighted average basis, effective July 1, 2013. By 2016, the competitive bidding process must either be rolled-out nationally or CMS must reduce Medicare prices in non-competitive bidding areas to match competitive bidding prices. We cannot predict at this time the full impact these changes and the developments in the competitive bidding program will have on our business and financial condition.
- 21 -
Table of Contents
Healthcare reform, including recently enacted legislation, may have a material adverse effect on our industry and our results of operations. In January 2011, the FDA announced 25 specific action items it intends to take with respect to the 510(k) process. The FDA issued its recommendations and proposed action items in response to concerns from both within and outside of the FDA about the 510(k) program. Although the FDA has not detailed the specific modifications or clarifications that the FDA intends to make to its guidance, policies, and regulations pertaining to the review and regulation of devices such as ours which seek and receive marketing clearance through the 510(k) process, the FDAs announced action items signal that additional regulatory requirements are likely. In particular, the FDA intends to issue a variety of draft guidance and regulations over the coming months which would, among other things, clarify when changes to a cleared medical device warrant a new 510(k) and which modifications would be eligible for a Special 510(k), establish a Unique Device Identification System, and clarify the FDAs use and application of several key terms in the 510(k) review process. These reforms, when implemented, could impose additional regulatory requirements upon us which could delay our ability to obtain new clearances, increase the costs of compliance, or restrict our ability to maintain our current clearances.
In March 2010, the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act (collectively, the PPACA) was signed into law in the United States. The PPACA makes changes that are expected to impact the medical device industry. One of the principal purposes of the PPACA was to expand health insurance coverage to approximately 32 million Americans who were uninsured. The PPACA requires adults not covered by an employer- or government-sponsored insurance plan to maintain health insurance coverage or pay a penalty, a provision commonly referred to as the individual mandate. We cannot predict the impact of these coverage expansions, if any, on the sales of our products.
The PPACA also contains a number of provisions designed to generate the revenues necessary to fund the coverage expansions. This includes new fees or taxes on certain health-related industries, including medical device manufacturers. Beginning in 2013, entities that manufacture, produce or import medical devices were required to pay an excise tax in an amount equal to 2.3% of the price for which such devices are sold in the United States. This excise tax is applicable to our products that are primarily used in hospitals and sleep labs, which includes the ApneaLink, VPAP Tx, certain Respiratory Care and dental sleep products. The PPACA also includes, among other things, the expansion of the second round two of competitive bidding to a total of 91 CBAs, and, as mentioned above, by 2016, the process must be nationalized or prices in non-competitive bidding areas must be adjusted to match competitive bidding prices. Other PPACA provisions include demonstrations to develop organizations that are paid under a new payment methodology for voluntary coordination of care by groups of providers, such as physicians and hospitals, and the establishment of a new Patient-Centered Outcomes Research Institute to oversee, identify priorities in and conduct comparative clinical effectiveness research. The increased funding and focus on comparative clinical effectiveness research, which compares and evaluates the risks and benefits, clinical outcomes, effectiveness and appropriateness of products, may result in lower reimbursements by payers for our products and decreased profits to us.
Other federal legislative changes have been proposed and adopted since the PPACA was enacted. On August 2, 2011, the President signed into law the Budget Control Act of 2011, which, among other things, creates the Joint Select Committee on Deficit Reduction to recommend proposals in spending reductions to Congress. The Joint Select Committee did not achieve a targeted deficit reduction of at least $1.2 trillion for the years 2013 through 2021, triggering the legislations automatic reduction to several government programs. This includes aggregate reductions to Medicare payments to providers, including home healthcare companies, of up to 2% per fiscal year. On January 2, 2013, President Obama signed into law the American Taxpayer Relief Act of 2012, or the ATRA, which delayed for another two months the budget cuts mandated by these sequestration provisions of the Budget Control
- 22 -
Table of Contents
Act of 2011. On March 1, 2013, the President signed an executive order implementing sequestration, and on April 1, 2013, the 2% Medicare payment reductions went into effect. The ATRA also, among other things, reduced Medicare payments to several providers, including hospitals, imaging centers and cancer treatment centers, and increased the statute of limitations period for the government to recover overpayments to providers from three to five years.
Various healthcare reform proposals have also emerged at the state level within the United States.
The PPACA as well as other federal and/or state healthcare reform measures that may be adopted in the future, singularly or in the aggregate, could have a material adverse effect on our business, financial condition and results of operations.
Failure to comply with anti-kickback and fraud regulations could result in substantial penalties and changes in our business operations. In particular, the U.S. Anti-Kickback Law prohibits persons from knowingly and willfully soliciting, receiving, offering or providing remuneration, directly or indirectly, to induce either the referral of an individual or the furnishing, recommending or arranging for a good or service, for which payment may be made under a federal healthcare program such as the Medicare and Medicaid programs. The U.S. government has interpreted this law broadly to apply to the marketing and sales activities of manufacturers and distributors like us.
The recently enacted PPACA, among other things, amends the intent requirement of the federal anti-kickback and criminal healthcare fraud statutes. A person or entity no longer needs to have actual knowledge of this statute or specific intent to violate it. In addition, the PPACA provides that the government may assert that a claim including items or services resulting from a violation of the federal anti-kickback statute constitutes a false or fraudulent claim for purposes of the false claims statutes. Many states and other governments have adopted laws similar to the federal Anti-Kickback Law. We are also subject to other federal and state fraud laws applicable to payment from any third-party payer. These laws prohibit persons from knowingly and willfully filing false claims or executing a scheme to defraud any healthcare benefit program, including private third-party payers. These laws may apply to manufacturers and distributors who provide information on coverage, coding, and reimbursement of their products to persons who do bill third-party payers. Any violation of these laws and regulations could result in civil and criminal penalties (including fines), increased legal expenses and exclusions from governmental reimbursement programs, all of which could have a material adverse effect upon our business, financial conditions and results of operations.
Beginning in 2013, the PPACA also imposes new reporting and disclosure requirements on device and drug manufacturers for any transfer of value made or distributed to prescribers and other healthcare providers. Device and drug manufacturers will also be required to report and disclose any investment interests held by physicians and their immediate family members during the preceding calendar year. Failure to submit required information may result in civil monetary penalties of up to an aggregate of $150,000 per year (and up to an aggregate of $1 million per year for knowing failures), for all payments, transfers of value or ownership or investment interests not reported in an annual submission. As of August 1, 2013, manufacturers are required to collect data, and they will be required to submit their first data reports to CMS by March 31, 2014 and by the 90th day of each calendar year thereafter.
In addition, there has been a recent trend of increased federal and state regulation of payments made to physicians for marketing. Some states, such as California, Massachusetts and Vermont, mandate implementation of compliance programs and/or the tracking and reporting of gifts, compensation and other remuneration to physicians. The shifting compliance environment and the need to build and maintain robust and expandable systems to comply with different compliance and/or reporting requirements in multiple jurisdictions increase the possibility that a healthcare company many run
- 23 -
Table of Contents
afoul of one or more of the requirements. The scope and enforcement of these laws are uncertain and subject to rapid change in the current environment of healthcare reform, especially in light of the lack of applicable precedent and regulations. Federal or state regulatory authorities might challenge our current or future activities under these laws. Any such challenge could have a material adverse effect on our business, results of operations and financial condition.
Complying with Food and Drug Administration, or FDA, and other regulations is an expensive and time-consuming process, and any failure to comply could have a materially adverse effect on our business, financial condition, or results of operations. We are subject to various U.S. federal, state, local and international regulations regarding our business activities. Failure to comply with these regulations could result in, among other things, recalls of our products, substantial fines and criminal charges against us or against our employees. Furthermore, our products could be subject to recall if the FDA, other regulators or we determine, for any reason, that our products are not safe or effective. Any recall or other regulatory action could increase our costs, damage our reputation, affect our ability to supply customers with the quantity of products they require and materially affect our operating results.
Product sales, introductions or modifications may be delayed or canceled as a result of FDA regulations or similar foreign regulations, which could cause our sales and profits to decline. Before we can market or sell a new medical device in the United States, we must obtain FDA clearance, which can be a lengthy and time-consuming process. We generally receive clearance from the FDA to market our products in the United States under Section 510(k) of the Federal Food, Drug, and Cosmetic Act or our products are exempt from the Section 510(k) clearance process. In the 510(k) clearance process, the FDA must determine that a proposed device is substantially equivalent to a device legally on the market, known as a predicate device, with respect to intended use, technology and safety and effectiveness, in order to clear the proposed device for marketing. The FDA has a high degree of latitude when evaluating submissions and may determine that a proposed device submitted for 510(k) clearance is not substantially equivalent to a predicate device. After a device receives 510(k) premarket notification clearance from the FDA, any modification that could significantly affect its safety or effectiveness, or that would constitute a major change in the intended use of the device, technology, materials, packaging, and certain manufacturing processes may require a new 510(k) clearance or premarket approval. We have modified some of our Section 510(k) approved products without submitting new Section 510(k) notices, which we do not believe were required. However, if the FDA disagrees with us and requires us to submit new Section 510(k) notifications for modifications to our existing products, we may be required to stop marketing the products while the FDA reviews the Section 510(k) notification.
Any new product introduction or existing product modification could be subjected to a lengthier, more rigorous FDA examination process. For example, in certain cases we may need to conduct clinical trials of a new product before submitting a 510(k) notice. We may also be required to obtain premarket approvals for certain of our products. Indeed, recent trends in the FDAs review of premarket notification submissions suggest that the FDA is often requiring manufacturers to provide new, more expansive, or different information regarding a particular device than what the manufacturer anticipated upon 510(k) submission. This has resulted in increasing uncertainty and delay in the premarket notification review process. In January 2011, the FDA announced twenty-five specific action items it intends to take with respect to the 510(k) process designed, in part, to provide greater transparency and certainty to the review process. Some of the changes that the FDA has announced it intends to take, may affect requirements related to which devices are eligible for Section 510(k) clearance and which devices may be used as predicates in demonstrating substantial equivalence, and the grounds and procedures under which the FDA may rescind a Section 510(k) clearance. Furthermore, the FDAs ongoing review of the 510(k) program may make it more difficult for us to make modifications to our previously cleared products, either by imposing more strict
- 24 -
Table of Contents
requirements on when a manufacturer must submit a new 510(k) for a modification to a previously cleared product, or by applying more onerous review criteria to such submissions. Specifically, on July 9, 2012, the FDA Safety and Innovation Act of 2012 was enacted, which, among other requirements, obligates the FDA to prepare a report for Congress on the FDAs approach for determining when a new 510(k) will be required for modifications or changes to a previously cleared device. After submitting this report, the FDA is expected to issue revised guidance to assist device manufacturers in making this determination. Until then, manufacturers may continue to adhere to the FDAs existing guidance on this topic when making a determination as to whether or not a new 510(k) is required for a change or modification to a device, but the practical impact of the FDAs continuing scrutiny of these issues remains unclear. These and other revisions to the FDAs 510(k) clearance process, when fully implemented, could impose additional regulatory requirements upon us that could delay our ability to obtain new clearances, increase the costs of compliance, or restrict our ability to maintain current clearances. The requirements of the more rigorous premarket approval process and/or significant changes to the Section 510(k) clearance process could delay product introductions and increase the costs associated with FDA compliance. Marketing and sale of our products outside the United States are also subject to regulatory clearances and approvals, and if we fail to obtain these regulatory approvals, our sales could suffer. We cannot assure you that any new products we develop will receive required regulatory approvals from U.S. or foreign regulatory agencies.
We are subject to substantial regulation related to quality standards applicable to our manufacturing and quality processes. Our failure to comply with these standards could have an adverse effect on our business, financial condition, or results of operations. The FDA regulates the approval, manufacturing, and sales and marketing of many of our products in the U.S. Significant government regulation also exists in Canada, Japan, Europe, and other countries in which we conduct business. As a device manufacturer, we are required to register with the FDA and are subject to periodic inspection by the FDA for compliance with the FDAs Quality System Regulation requirements, which require manufacturers of medical devices to adhere to certain regulations, including testing, quality control and documentation procedures. In addition, the federal Medical Device Reporting regulations require us to provide information to the FDA whenever there is evidence that reasonably suggests that a device may have caused or contributed to a death or serious injury or, if a malfunction were to occur, could cause or contribute to a death or serious injury. Compliance with applicable regulatory requirements is subject to continual review and is rigorously monitored through periodic inspections by the FDA. In the European Community, we are required to maintain certain ISO certifications in order to sell our products and must undergo periodic inspections by notified bodies to obtain and maintain these certifications. Failure to comply with current governmental regulations and quality assurance guidelines could lead to temporary manufacturing shutdowns, product recalls or related field actions, product shortages or delays in product manufacturing. Efficacy or safety concerns, an increase in trends of adverse events in the marketplace, and/or manufacturing quality issues with respect to our products could lead to product recalls or related field actions, withdrawals, and/or declining sales.
Off-label marketing of our products could result in substantial penalties. Clearance under Section 510(k) only permits us to market our products for the uses indicated on the labeling cleared by the FDA. We may request additional label indications for our current products, and the FDA may deny those requests outright, require additional expensive clinical data to support any additional indications or impose limitations on the intended use of any cleared products as a condition of clearance. If the FDA determines that we have marketed our products for off-label use, we could be subject to fines, injunctions or other penalties.
Disruptions in the supply of components from our single source suppliers could result in a significant reduction in sales and profitability. We purchase uniquely configured components for
- 25 -
Table of Contents
our devices from various suppliers, including some who are single-source suppliers for us. We cannot assure you that a replacement supplier would be able to configure its components for our devices on a timely basis or, in the alternative, that we would be able to reconfigure our devices to integrate the replacement part. A reduction or halt in supply while a replacement supplier reconfigures its components, or while we reconfigure our devices for the replacement part, would limit our ability to manufacture our devices, which could result in a significant reduction in sales and profitability. We cannot assure you that our inventories would be adequate to meet our production needs during any prolonged interruption of supply.
We are subject to potential product liability claims that may exceed the scope and amount of our insurance coverage, which would expose us to liability for uninsured claims. We are subject to potential product liability claims as a result of the design, manufacture and marketing of medical devices. Any product liability claim brought against us, with or without merit, could result in the increase of our product liability insurance rates. In addition, we would have to pay any amount awarded by a court in excess of our policy limits. Our insurance policies have various exclusions, and thus we may be subject to a product liability claim for which we have no insurance coverage, in which case, we may have to pay the entire amount of any award. We cannot assure you that our insurance coverage will be adequate or that all claims brought against us will be covered by our insurance and we cannot assure you that we will be able to obtain insurance in the future on terms acceptable to us or at all. A successful product liability claim brought against us in excess of our insurance coverage, if any, may require us to pay substantial amounts, which could harm our business.
Our intellectual property may not protect our products, and/or our products may infringe on the intellectual property rights of third-parties. We rely on a combination of patents, trade secrets and non-disclosure agreements to protect our intellectual property. Our success depends, in part, on our ability to obtain and maintain United States and foreign patent protection for our products, their uses and our processes to preserve our trade secrets and to operate without infringing on the proprietary rights of third-parties. We have a number of pending patent applications, and we do not know whether any patents will issue from any of these applications. We do not know whether any of the claims in our issued patents or pending applications will provide us with any significant protection against competitive products or otherwise be commercially valuable. Legal standards regarding the validity of patents and the proper scope of their claims are still evolving, and there is no consistent law or policy regarding the valid breadth of claims. Additionally, there may be third-party patents, patent applications and other intellectual property relevant to our products and technology which are not known to us and that block or compete with our products.
We face the risks that:
| | third-parties will infringe our intellectual property rights; |
| | our non-disclosure agreements will be breached; |
| | we will not have adequate remedies for infringement; |
| | our trade secrets will become known to or independently developed by our competitors; or |
| | third-parties will be issued patents that may prevent the sale of our products or require us to license and pay fees or royalties in order for us to be able to market some of our products. |
Litigation may be necessary to enforce patents issued to us, to protect our proprietary rights, or to defend third-party claims that we have infringed upon proprietary rights of others. The defense and prosecution of patent claims, including these pending claims, as well as participation in other inter-party proceedings, can be expensive and time-consuming, even in those instances in which the outcome is favorable to us. If the outcome of any litigation or proceeding brought against us were adverse, we could be subject to significant liabilities to third-parties, could be required to obtain
- 26 -
Table of Contents
licenses from third-parties, could be forced to design around the patents at issue or could be required to cease sales of the affected products. A license may not be available at all or on commercially viable terms, and we may not be able to redesign our products to avoid infringement. Additionally, the laws regarding the enforceability of patents vary from country to country, and we cannot assure you that any patent issues we face will be uniformly resolved, or that local laws will provide us with consistent rights and benefits.
We are subject to tax audits by various tax authorities in many jurisdictions. From time to time we may be audited by tax authorities in various jurisdictions. Any final assessment resulting from such audits could result in material changes to our past or future taxable income, tax payable or deferred tax assets, and could require us to pay penalties and interest that could materially adversely affect our financial results.
Our quarterly operating results are subject to fluctuation for a variety of reasons. Our operating results have, from time to time, fluctuated on a quarterly basis and may be subject to similar fluctuations in the future. These fluctuations may result from a number of factors, including:
| | the introduction of new products by us or our competitors; |
| | the geographic mix of product sales; |
| | the success and costs of our marketing efforts in new regions; |
| | changes in third-party payer reimbursement; |
| | timing of regulatory clearances and approvals; |
| | timing of orders by distributors; |
| | expenditures incurred for research and development; |
| | competitive pricing in different regions; |
| | the effect of foreign currency transaction gains or losses; and |
| | other activities of our competitors. |
Fluctuations in our quarterly operating results may cause the market price of our common stock to fluctuate.
If a natural or man-made disaster strikes our manufacturing facilities, we will be unable to manufacture our products for a substantial amount of time and our sales and profitability will decline. Our facilities and the manufacturing equipment we use to produce our products would be costly to replace and could require substantial lead-time to repair or replace. The facilities may be affected by natural or man-made disasters and in the event they were affected by a disaster, we would be forced to rely on third-party manufacturers. Although we believe we possess adequate insurance for the disruption of our business from causalities, such insurance may not be sufficient to cover all of our potential losses and may not continue to be available to us on acceptable terms, or at all.
Delaware law and provisions in our charter and could make it difficult for another company to acquire us. Provisions of our certificate of incorporation may have the effect of delaying or preventing changes in control or management which might be beneficial to us or our security holders. In particular, our board of directors is divided into three classes, serving for staggered three-year terms. Because of this classification, it will require at least two annual meetings to elect directors constituting a majority of our board of directors. Additionally, our board of directors has the authority to issue up to 2,000,000 shares of preferred stock and to determine the price, rights, preferences, privileges and restrictions, including voting rights, of those shares without further vote or action by
- 27 -
Table of Contents
the stockholders. The rights of the holders of our common stock will be subject to, and may be adversely affected by, the rights of the holders of any preferred stock that may be issued in the future. The issuance of preferred stock may have the effect of delaying, deferring or preventing a change in control, may discourage bids for our common stock at a premium over the market price of our common stock and may adversely affect the market price of our common stock and the voting and other rights of the holders of our common stock.
You may not be able to enforce the judgments of U.S. courts against some of our assets or officers and directors. A substantial portion of our assets are located outside the United States. Additionally, some of our directors and executive officers reside outside the United States, along with all or a substantial portion of their assets. As a result, it may not be possible for investors to enforce judgments of U.S. courts relating to any liabilities under U.S. securities laws against our assets, those persons or their assets. In addition, investors may not be able to pursue claims based on U.S. securities laws against these assets or these persons in Australian courts, where most of these assets and persons reside.
Our results of operations may be materially affected by global economic conditions generally, including conditions in the financial markets. Recently, concerns over inflation, energy costs, geopolitical issues, the availability and cost of credit, the United States mortgage market, a declining residential real estate market in the United States, and the ability of sovereign nations to pay their debts have contributed to increased volatility and diminished expectations for the economy and the financial markets going forward. These factors, combined with volatile commodity prices, declining business and consumer confidence and increased unemployment, have precipitated an economic slowdown. It is difficult to predict how long the current economic conditions will continue and whether the economic conditions will continue to deteriorate. If the economic climate in the United States or outside the United States continues to deteriorate or there is a shift in government spending priorities, customers or potential customers could reduce or delay their purchases, which could impact our revenue, our ability to manage inventory levels, collect customer receivables, and ultimately decrease our profitability.
| ITEM 1B | UNRESOLVED STAFF COMMENTS |
We have received no written comments regarding our periodic or current reports from the staff of the Securities and Exchange Commission that were issued 180 days or more preceding the end of our fiscal year 2013 that remain unresolved.
| ITEM 2 | PROPERTIES |
We conduct our operations in both owned and leased properties. Our principal executive offices and U.S. sales facilities, consisting of approximately 230,000 square feet, are located on Spectrum Center Boulevard in San Diego, California, in a building we own. We have our research and development and office facilities and our principal manufacturing facility at our owned site in Norwest, Sydney, Australia. Sales and warehousing facilities are leased in South Carolina, U.S.A.; Abingdon, England; Munich, Bremen, Hochstadt, Germany; Lyon, Paris, France; Basel, Switzerland; Stockholm, Sweden; Helsinki, Finland; Oslo, Norway; New Delhi, India; Tokyo, Japan; Dublin, Ireland and Kowloon, Hong Kong.
- 28 -
Table of Contents
We believe that our facilities are adequate to meet the needs of our current business operations. At June 30, 2013, our principal owned and leased properties were as follows:
| Location | Ownership Status (Owned / Leased) |
Square footage | Primary Usage | |||||
| San Diego, California |
Owned | 230,000 | Corporate headquarters, sales and administration | |||||
| Norwest, Sydney, Australia |
Owned | 224,000 | Principal manufacturing facility, engineering, research and development | |||||
| Chatsworth, California |
Leased | 72,000 | Motor manufacturing, engineering, research and development | |||||
| Duncan, South Carolina |
Leased | 174,000 | Manufacturing, warehouse and distribution | |||||
| Moreno Valley, California |
Leased | 130,000 | Warehouse and distribution | |||||
| Singapore, Singapore |
Leased | 95,000 | Manufacturing facility | |||||
| Munich, Germany |
Leased | 119,000 | Sales and distribution, research and development | |||||
| Lyon, France | Leased | 52,000 | Sales and distribution | |||||
| Paris, France | Leased | 43,000 | Manufacturing facility, field service | |||||
| Freudenstadt, Germany |
Owned | 43,000 | Manufacturing facility | |||||
| Johor Bahru, Malaysia |
Leased | 46,000 | Manufacturing facility | |||||
| ITEM 3 | LEGAL PROCEEDINGS |
See Note 19 Legal Actions and Contingencies to the consolidated financial statements for a summary of legal proceedings.
| ITEM 4 | MINE SAFETY DISCLOSURES |
Not Applicable.
- 29 -
Table of Contents
PART II
| ITEM 5 | MARKET FOR REGISTRANTS COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES |
Our common stock is traded on the NYSE under the symbol RMD. The following table sets forth for the fiscal periods indicated the high and low closing prices for the common stock as reported by the NYSE.
| 2013 | 2012 | |||||||||||||||
| High | Low | High | Low | |||||||||||||
| Quarter One, Ended September 30 |
$ | 40.47 | $ | 30.63 | $ | 33.17 | $ | 25.76 | ||||||||
| Quarter Two, Ended December 31 |
42.77 | 38.82 | 31.70 | 23.46 | ||||||||||||
| Quarter Three, Ended March 31 |
48.37 | 42.36 | 31.99 | 24.81 | ||||||||||||
| Quarter Four, Ended June 30 |
51.17 | 44.02 | 35.01 | 29.84 | ||||||||||||
At August 5, 2013, there were 24 holders of record of our common stock, although many of these holders of record own shares as nominees on behalf of other beneficial owners. During fiscal years 2013 and 2012, we paid dividends totaling $97.2 million and Nil, respectively. On August 1, 2013, the directors declared an increased quarterly dividend from $0.17 per share to $0.25 per share, which has a record date of August 20, 2013 and will be payable on September 17, 2013. We pay the dividend in U.S. currency to holders of our common stock trading on the NYSE. Holders of CDIs trading on the ASX will receive an equivalent amount in Australian currency based on the exchange rate on August 20, 2013 and reflecting the 10:1 ratio between CDIs and NYSE shares. The ex-dividend rate date will be August 14, 2013 for CDI holders and August 16, 2013 for holders of our common stock. As a result of these differences, we have received a waiver from the ASXs settlement operating rules, which will allow us to defer processing conversions between its common stock and CDI registers from August 14, 2013 through August 20, 2013, inclusive. We expect the dividend will continue to be unfranked for Australian tax purposes. We expect to fund our dividend commitments with our operating cash flows and existing loan facilities.
Securities Authorized for Issuance Under Equity Compensation Plans
The information included under Item 12 of Part III of this Report, Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters, is hereby incorporated by reference into this Item 5 of Part II of this Report.
- 30 -
Table of Contents
Purchases of Equity Securities
The following table summarizes purchases by us of our common stock during the fiscal year ending June 30, 2013:
| Period | Total Number of Shares Purchased |
Average Price Paid per Share |
Total Number of Shares Purchased as Part of Publicly Announced Programs(1) |
Maximum Number of Shares that May Yet Be Purchased Under the Programs(1) |
||||||||||||
| July 2012 |
0 | $ | 0 | 27,731,749 | 8,843,432 | |||||||||||
| August 2012 |
215,863 | 37.50 | 27,947,612 | 8,627,569 | ||||||||||||
| September 2012 |
0 | 0 | 27,947,612 | 8,627,569 | ||||||||||||
| October 2012 |
100,000 | 40.01 | 28,047,612 | 8,527,569 | ||||||||||||
| November 2012 |
841,530 | 39.90 | 28,889,142 | 7,686,039 | ||||||||||||
| December 2012 |
58,470 | 40.71 | 28,947,612 | 7,627,569 | ||||||||||||
| January 2013 |
52,500 | 43.91 | 29,000,112 | 7,575,069 | ||||||||||||
| February 2013 |
924,328 | 43.68 | 29,924,440 | 6,650,741 | ||||||||||||
| March 2013 |
561,977 | 43.79 | 30,486,417 | 6,088,764 | ||||||||||||
| April 2013 |
226,470 | 44.33 | 30,712,887 | 5,862,294 | ||||||||||||
| May 2013 |
363,126 | 48.92 | 31,076,013 | 5,499,168 | ||||||||||||
| June 2013 |
950,000 | 47.24 | 32,026,013 | 4,549,168 | ||||||||||||
| Total |
4,294,264 | $ | 43.78 | 32,026,013 | 4,549,168 | |||||||||||
(1) On August 24, 2011, our board of directors approved a new share repurchase program, authorizing us to acquire up to an aggregate of 20.0 million shares of ResMed Inc. common stock. The program allows us to repurchase shares of our common stock from time to time for cash in the open market, or in negotiated or block transactions, as market and business conditions warrant. The program authorizes us to purchase in addition to the shares we repurchased under our previous programs. There is no expiration date for this program. All share repurchases since August 24, 2011 have been executed in accordance with this program. Since the inception of the share repurchase programs, we have repurchased 32.0 million shares at a total cost of $1.1 billion.
- 31 -
Table of Contents
PERFORMANCE GRAPH
This performance graph is furnished and shall not be deemed filed with the SEC or subject to Section 18 of the Exchange Act, nor shall it be deemed incorporated by reference in any of our filings under the Securities Act of 1933, as amended.
The following graph compares the cumulative total stockholders return on our common stock from June 30, 2008 through June 30, 2013, with the comparable cumulative return of the S&P 500 index, the S&P 500 Health Care index, and the Dow Jones US Medical Devices index. The graph assumes that $100 was invested in our common stock and each index on June 30, 2008. In addition, the graph assumes the reinvestment of all dividends paid. The stock price performance on the following graph is not necessarily indicative of future stock price performance.
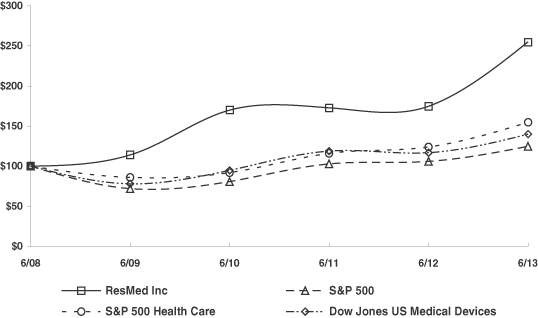
The following table shows total indexed return of stock price plus reinvestments of dividends, assuming an initial investment of $100 at June 30, 2008, for the indicated periods.
| Index | June 2008 | June 2009 | June 2010 | June 2011 | June 2012 | June 2013 | ||||||||||||||||||
| ResMed Inc |
100 | 114 | 170 | 173 | 175 | 255 | ||||||||||||||||||
| S&P 500 |
100 | 72 | 81 | 103 | 106 | 125 | ||||||||||||||||||
| S&P 500 Health Care |
100 | 86 | 92 | 116 | 124 | 155 | ||||||||||||||||||
| Dow Jones US Medical Devices |
100 | 78 | 95 | 119 | 117 | 140 | ||||||||||||||||||
- 32 -
Table of Contents
| ITEM 6 | SELECTED FINANCIAL DATA |
The following table summarizes certain selected consolidated financial data for, and as of the end of, each of the fiscal years in the five-year period ended June 30, 2013. The data set forth below should be read in conjunction with Item 7 of Part II of this Report, Managements Discussion and Analysis of Financial Condition and Results of Operations and Item 8 of Part II of this Report, Consolidated Financial Statements and Supplementary Data, and related Notes included elsewhere in this Report. The consolidated statements of operations data for the years ended June 30, 2013, 2012 and 2011 and the balance sheet data as of June 30, 2013 and 2012 are derived from our audited consolidated financial statements included elsewhere in this Report. The consolidated statements of operations data for the years ended June 30, 2010 and 2009 and the balance sheet data as of June 30, 2011, 2010 and 2009 are derived from our audited consolidated financial statements not included herein. Historical results are not necessarily indicative of the results to be expected in the future, and the results for the years presented should not be considered indicative of our future results of operations.
| Consolidated Statement of Income Data | Years Ended June 30, | |||||||||||||||||||
| (In thousands, except per share data): | 2013 | 2012 | 2011 | 2010 | 2009 | |||||||||||||||
| Net revenues |
$ | 1,514,457 | $ | 1,368,515 | $ | 1,243,148 | $ | 1,092,357 | $ | 920,735 | ||||||||||
| Cost of sales |
573,800 | 547,780 | 501,822 | 436,874 | 366,933 | |||||||||||||||
| Gross profit |
940,657 | 820,735 | 741,326 | 655,483 | 553,802 | |||||||||||||||
| Selling, general and administrative expenses | 430,802 | 402,621 | 372,249 | 331,858 | 293,375 | |||||||||||||||
| Research and development expenses | 120,124 | 109,733 | 92,007 | 75,202 | 63,056 | |||||||||||||||
| Education, research and settlement charge | 24,765 | - | - | - | - | |||||||||||||||
| Amortization of acquired intangible assets | 10,142 | 13,974 | 10,146 | 8,041 | 7,060 | |||||||||||||||
| Total operating expenses |
585,833 | 526,328 | 474,402 | 415,101 | 363,491 | |||||||||||||||
| Income from operations |
354,824 | 294,407 | 266,924 | 240,382 | 190,311 | |||||||||||||||
| Other income: |
||||||||||||||||||||
| Interest income, net |
32,486 | 29,080 | 26,043 | 14,029 | 10,205 | |||||||||||||||
| Other, net |
(2,191 | ) | 8,458 | 10,740 | 6,178 | 1,168 | ||||||||||||||
| Total other income, net |
30,295 | 37,538 | 36,783 | 20,207 | 11,373 | |||||||||||||||
| Income before income taxes |
385,119 | 331,945 | 303,707 | 260,589 | 201,684 | |||||||||||||||
| Income taxes |
(77,986 | ) | (77,095 | ) | (76,721 | ) | (70,504 | ) | (55,236 | ) | ||||||||||
| Net income |
$ | 307,133 | $ | 254,850 | $ | 226,986 | $ | 190,085 | $ | 146,448 | ||||||||||
| Basic earnings per share |
$ | 2.15 | $ | 1.75 | $ | 1.49 | $ | 1.26 | $ | 0.97 | ||||||||||
| Diluted earnings per share |
$ | 2.10 | $ | 1.71 | $ | 1.44 | $ | 1.23 | $ | 0.95 | ||||||||||
| Dividends per share |
$ | 0.68 | $ | - | $ | - | $ | - | $ | - | ||||||||||
| Weighted average: |
||||||||||||||||||||
| Basic shares outstanding |
142,954 | 145,901 | 152,471 | 150,908 | 151,258 | |||||||||||||||
| Diluted shares outstanding |
146,410 | 149,316 | 157,195 | 155,098 | 154,226 | |||||||||||||||
- 33 -
Table of Contents
| Consolidated Balance Sheet Data | As of June 30, | |||||||||||||||||||
| (In thousands): | 2013 | 2012 | 2011 | 2010 | 2009 | |||||||||||||||
| Working capital |
$ | 874,800 | $ | 1,108,299 | $ | 1,083,612 | $ | 672,669 | $ | 584,184 | ||||||||||
| Total assets |
2,210,721 | 2,137,869 | 2,068,922 | 1,626,397 | 1,507,968 | |||||||||||||||
| Long-term debt, less current maturities |
769 | 250,783 | 100,000 | - | 94,191 | |||||||||||||||
| Total stockholders equity |
$ | 1,610,516 | $ | 1,607,627 | $ | 1,730,737 | $ | 1,287,536 | $ | 1,115,192 | ||||||||||
- 34 -
Table of Contents
| ITEM 7 | MANAGEMENTS DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
Overview
Managements discussion and analysis of financial condition and results of operations is intended to help the reader understand the results of operations and financial condition of ResMed Inc and subsidiaries. It is provided as a supplement to, and should be read in conjunction with the selected financial data and consolidated financial statements and notes included elsewhere in this Report.
We are a leading developer, manufacturer and distributor of medical equipment for treating, diagnosing, and managing sleep-disordered breathing (SDB) and other respiratory disorders. During the fiscal year, we continued our efforts to build awareness of the consequences of untreated SDB and to grow our business in this market. In our efforts, we have attempted to raise awareness through market and clinical initiatives and by highlighting the increasing link between the potential effects SDB can have on co-morbidities such as cardiac disease, diabetes, hypertension and obesity.
There are many studies being conducted that provide new evidence that treating SDB and OSA can improve health, quality of life and also mitigate the dangers of sleep apnea in occupational health and safety, especially in the transport industry. Evidence continues to mount supporting the role of SDB therapy for disease prevention, improvement of quality of life and healthcare cost reduction. The following three recent studies support the role of SDB therapy for disease prevention, improvement of quality of life and healthcare cost reduction:
| | In a ResMed-sponsored study published in Population Health Management involving 22,000 members on the Union Pacific Railroad health plan, findings suggest that a low-cost, patient-focused SDB education campaign can improve healthcare outcomes and reduce medical expenses. First, the study showed that members of the Union Pacific plan who had untreated SDB had higher medical expenses than employees without the disease and, second, it demonstrated that treatment of SDB with positive airway pressure (PAP) therapy reduced medical costs, in-patient costs and hospital admissions. After the campaign was initiated, the healthcare plan realized cost savings of $4.9 million over a two-year period. |
| | A study published in the June 2012 issue of the American Journal of Managed Care demonstrated that newly diagnosed SDB patients who initiated PAP therapy had significantly lower hospitalization risk and lower all-cause healthcare costs compared to patients who did not use PAP. |
| | In the July 2013 issue of the Journal of Cardiac Failure, a study showed that central sleep apnea and severe obstructive sleep apnea are independent risk factors for six-month cardiac hospital readmission. |
We are committed to ongoing investment in research and development and product enhancements. During fiscal year 2013, we invested approximately $120.1 million on research and development activities, which represents approximately 8% of net revenues. Since the development of CPAP, we have developed a number of innovative products for the treatment of SDB and other respiratory disorders including airflow generators, diagnostic products, mask systems, headgear and other accessories. During fiscal year 2013, we released new products across both our mask and flow generator categories, including the VPAP COPD, Quattro Air, Swift FX Bella, Swift FX Nano and ResMeds SleepSeeker. The release of these products as well as the release of the S9 bilevel range of flow generators, and Stellar 100 and 150 ventilation devices in fiscal 2011, have all contributed to the increase in our net revenues for fiscal year 2013.
We reported record financial results in fiscal year 2013, with an increase in net revenue to $1,514.5 million, an increase of 11% when compared to fiscal year 2012. Gross profit increased for the year
- 35 -
Table of Contents
ended June 30, 2013 to $940.7 million, from $820.7 million for the year ended June 30, 2012, an increase of $119.9 million or 15%. Our net income for the year ended June 30, 2013 was $307.1 million or $2.10 per diluted share compared to net income of $254.9 million or $1.71 per diluted share for the year ended June 30, 2012.
Total operating cash flow for fiscal year 2013 was $402.8 million and at June 30, 2013, our cash and cash equivalents totaled $876.0 million. Our total assets increased by 3% to $2.2 billion and our shareholders equity was $1.6 billion. During fiscal year 2013, we repurchased 4.3 million shares at a cost of $188.0 million under our share repurchase program, compared to 13.6 million shares at a cost of $391.2 million during fiscal year 2012. We initiated a quarterly dividend of $0.17 per share during fiscal 2013 with a total amount of $97.2 million paid to stockholders.
In order to provide a framework for assessing how our underlying businesses performed, excluding the effect of foreign currency fluctuations, we provide certain financial information on a constant currency basis, which is in addition to the actual financial information presented. In order to calculate our constant currency information, we translate the current period financial information using the foreign currency exchange rates that were in effect during the previous comparable period. However, constant currency measures should not be considered in isolation or as an alternative to U.S. dollar measures that reflect current period exchange rates, or to other financial measures calculated and presented in accordance with U.S. generally accepted accounting principles.
Fiscal Year Ended June 30, 2013 Compared to Fiscal Year Ended June 30, 2012
Net Revenues. Net revenue increased for the year ended June 30, 2013 to $1,514.5 million from $1,368.5 million for the year ended June 30, 2012, an increase of $145.9 million or 11%. The increase in net revenue was attributable to an increase in unit sales of our flow generators, masks and accessories. Movements in international currencies against the U.S. dollar negatively impacted revenues by approximately $15.1 million for the year ended June 30, 2013. Excluding the impact of unfavorable foreign currency movements, sales for the year ended June 30, 2013 increased by 12% compared to the year ended June 30, 2012.
Net revenue in North and Latin America increased for the year ended June 30, 2013 to $851.6 million from $749.0 million for the year ended June 30, 2012, an increase of $102.6 million or 14%. We believe this increase primarily reflects growth in the overall SDB market and market share gains in the APAP and bilevel flow generator segments.
Net revenue in markets outside North and Latin America increased for the year ended June 30, 2013 to $662.8 million from $619.5 million for the year ended June 30, 2012, an increase of $43.4 million or 7%. Excluding the impact of unfavorable foreign currency movements, international sales for the year ended June 30, 2013 increased by 9%, compared to the year ended June 30, 2012. We believe this increase in sales outside North and Latin America predominantly reflects growth in the overall SDB market.
Net revenue from flow generators for the year ended June 30, 2013 totaled $823.5 million from $736.6 million for the year ended June 30, 2012, an increase of 12%, including increases of 18% in North and Latin America and 7% elsewhere. Net revenue from mask systems, motors and other accessories totaled $690.9 million, an increase of 9%, including increases of 11% in North and Latin America and 7% elsewhere, for the year ended June 30, 2013, compared to the year ended June 30, 2012. We believe these increases primarily reflect growth in the overall SDB market and market share gains in the APAP and bilevel flow generator segments.
- 36 -
Table of Contents
The following table summarizes the percentage movements in our net revenue for the year ended June 30, 2013 compared to the year ended June 30, 2012:
| North and Latin America |
International | Total | International (Constant Currency)* |
Total (Constant Currency)* |
||||||||||||||||
| Flow generators |
18 | % | 7 | % | 12 | % | 9 | % | 13 | % | ||||||||||
| Masks and other accessories |
11 | % | 7 | % | 9 | % | 10 | % | 10 | % | ||||||||||
|
Total |
14 | % | 7 | % | 11 | % | 9 | % | 12 | % | ||||||||||
* Constant currency numbers exclude the impact of movements in international currencies.
Gross Profit. Gross profit increased for the year ended June 30, 2013 to $940.7 million from $820.7 million for the year ended June 30, 2012, an increase of $119.9 million or 15%. Gross profit as a percentage of net revenue was 62.1% for the year ended June 30, 2013, compared with the 60.0% for the year ended June 30, 2012. The improvement in gross margins was primarily due to cost savings attributable to manufacturing and supply chain improvements, and favorable change in product mix as sales of our higher margin products represented a higher proportion of our sales, partially offset by declines in our average selling prices.
Selling, General and Administrative Expenses. Selling, general and administrative expenses increased for the year ended June 30, 2013 to $430.8 million from $402.6 million for the year ended June 30, 2012, an increase of $28.2 million or 7%. As a percentage of net revenue, selling, general and administrative expenses for the year ended June 30, 2013 was 28%, compared to 29% for the year ended June 30, 2012.
The increase in selling, general and administrative expenses was primarily due to an increase in the number of sales and administrative personnel to support our growth and other expenses related to the increase in our sales including activities targeted at increasing the awareness and diagnosis of SDB. As a percentage of net revenue, we expect our future selling, general and administrative expense to be around 28%.
Research and Development Expenses. Research and development expenses increased for the year ended June 30, 2013 to $120.1 million from $109.7 million for the year ended June 30, 2012, an increase of $10.4 million or 9%. As a percentage of net revenue, research and development expenses were 8% for the year ended June 30, 2013 compared to 8% for the year ended June 30, 2012.
The increase in research and development expenses was primarily due to an increase in the number of research and development personnel, consulting and contractor expenses and an increase in materials and tooling costs incurred to facilitate development of new products. We expect our future research and development expenses, as a percentage of revenue, to be in the range of 7% to 8%.
Education, research and settlement charge. During the year we agreed to pay the University of Sydney $24.8 million to establish two perpetual academic chairs, fund future research in the fields of sleep medicine and biomedical engineering, and settle legal proceedings between us. We have expensed the full amount of $24.8 million ($17.7 million, net of tax) in the current year within our operating expenses and we have separately disclosed the amount as an education, research and settlement charge.
Amortization of Acquired Intangible Assets. Amortization of acquired intangible assets for the year ended June 30, 2013 totaled $10.1 million compared to $14.0 million for the year ended June 30, 2012. The reduction in amortization expense is mainly attributable to certain acquired intangibles reaching the end of their useful life and therefore being fully amortized.
- 37 -
Table of Contents
Total other income, net. Total other income, net for the year ended June 30, 2013 was $30.3 million, a decrease of $7.2 million compared with $37.5 million for the year ended June 30, 2012. The decrease in total other income, net, was due primarily to losses on foreign currency transactions, partially offset by an increase in interest income, due primarily to an increase in cash balances held.
Income Taxes. Our effective income tax rate decreased to 20.2% for the year ended June 30, 2013 from 23.2% for the year ended June 30, 2012. The lower effective income tax rate was primarily due to a change in the geographic mix of our taxable income, including the lower statutory tax rates and other incentives associated with our Singapore and Malaysia manufacturing operations.
Net Income. As a result of the factors above and share repurchases, our net income and earnings per share for the year ended June 30, 2013 was $307.1 million or $2.10 per diluted share compared to net income of $254.9 million or $1.71 per diluted share for the year ended June 30, 2012, an increase of 21% and 23%, respectively, over the year ended June 30, 2012.
Fiscal Year Ended June 30, 2012 Compared to Fiscal Year Ended June 30, 2011
Net Revenues. Net revenue increased for the year ended June 30, 2012 to $1,368.5 million from $1,243.1 million for the year ended June 30, 2011, an increase of $125.4 million or 10%. The increase in net revenue was attributable to an increase in unit sales of our flow generators, masks and accessories. Movements in international currencies against the U.S. dollar negatively impacted revenues by approximately $7.3 million for the year ended June 30, 2012. Excluding the impact of unfavorable foreign currency movements, sales for the year ended June 30, 2012 increased by 11% compared to the year ended June 30, 2011.
Net revenue in North and Latin America increased for the year ended June 30, 2012 to $749.0 million from $662.2 million for the year ended June 30, 2011, an increase of $86.8 million or 13%. We believe this increase predominantly reflected growth in the overall SDB market and growth generated from our recent product releases including the S9 bilevel flow generators and the Quattro FX, Mirage FX and Mirage FX for Her masks.
Net revenue in markets outside North and Latin America increased for the year ended June 30, 2012 to $619.5 million from $580.9 million for the year ended June 30, 2011, an increase of $38.6 million or 7%. Excluding the impact of unfavorable foreign currency movements, international sales for the year ended June 30, 2012 increased by 8%, compared to the year ended June 30, 2011. We believe this increase in sales outside North and Latin America predominantly reflected growth in the overall SDB market and growth generated from our recent product releases including the S9 bilevel flow generators and the Quattro FX and Mirage FX masks.
Net revenue from flow generators for the year ended June 30, 2012 totaled $736.6 million from $699.3 million for the year ended June 30, 2011, an increase of 5%, including increases of 6% in North and Latin America and 5% elsewhere. Net revenue from mask systems, motors and other accessories totaled $631.9 million, an increase of 16%, including increases of 19% in North and Latin America and 11% elsewhere, for the year ended June 30, 2012, compared to the year ended June 30, 2011. We believe these increases primarily reflected growth in the overall SDB market and contributions from new products.
- 38 -
Table of Contents
The following table summarizes the percentage movements in our net revenue for the year ended June 30, 2012 compared to the year ended June 30, 2011:
| North and Latin America |
International | Total | International (Constant Currency)* |
Total (Constant Currency)* |
||||||||||||||||
| Flow generators |
6 | % | 5 | % | 5 | % | 6 | % | 6 | % | ||||||||||
| Masks and other accessories |
19 | % | 11 | % | 16 | % | 11 | % | 16 | % | ||||||||||
| Total |
13 | % | 7 | % | 10 | % | 8 | % | 11 | % | ||||||||||
* Constant currency numbers exclude the impact of movements in international currencies.
Gross Profit. Gross profit increased for the year ended June 30, 2012 to $820.7 million from $741.3 million for the year ended June 30, 2011, an increase of $79.4 million or 11%. Gross profit as a percentage of net revenue remained at 60% for the year ended June 30, 2012, which was consistent with the 60% for the year ended June 30, 2011. Gross margins were positively impacted by a favorable change in product mix as sales of our higher margin products represented a higher proportion of our sales and cost savings attributable to manufacturing and supply chain improvements. These impacts were offset by negative impacts associated with declines in our average selling prices and the appreciation of the Australian dollar against the U.S. dollar as the majority of our manufacturing labor and overhead was denominated in Australian dollars.
Selling, General and Administrative Expenses. Selling, general and administrative expenses increased for the year ended June 30, 2012 to $402.6 million from $372.2 million for the year ended June 30, 2011, an increase of $30.4 million or 8%. As a percentage of net revenue, selling, general and administrative expenses for the year ended June 30, 2012 was 29%, compared to 30% for the year ended June 30, 2011.
The increase in selling, general and administrative expenses was primarily due to an increase in the number of sales and administrative personnel to support our growth and other expenses related to the increase in our sales including activities targeted at increasing the awareness and diagnosis of SDB.
Research and Development Expenses. Research and development expenses increased for the year ended June 30, 2012 to $109.7 million from $92.0 million for the year ended June 30, 2011, an increase of $17.7 million or 19%.
The increase in research and development expenses was primarily due to an increase in the number of research and development personnel and an increase in clinical trial costs. The increase in research and development expenses was also due to the appreciation of international currencies against the U.S. dollar, which increased our research and development expenses by approximately $2.5 million for the year ended June 30, 2012, as reported in U.S. dollars. We expect our future research and development expenses, as a percentage of revenue, to be in the range of 8%.
Amortization of Acquired Intangible Assets. Amortization of acquired intangible assets for the year ended June 30, 2012 totaled $14.0 million compared to $10.1 million for the year ended June 30, 2011. The increase in amortization expense was attributable to our recent acquisitions of BiancaMed Limited and Gruendler GmbH in the first quarter of fiscal year 2012.
Other Income (Expense), Net. Other income, net for the year ended June 30, 2012 was $37.5 million, an increase of $0.8 million over $36.8 million for the year ended June 30, 2011. The increase in other income, net, was due to an increase in interest income, due primarily to an increase in cash balances held, partially offset by lower gains on foreign currency and hedging transactions and an increase in interest expense due to an increase in borrowings.
- 39 -
Table of Contents
Income Taxes. Our effective income tax rate decreased to 23.2% for the year ended June 30, 2012 from 25.3% for the year ended June 30, 2011. The lower tax rate was primarily due to the geographic mix of taxable income, including the impact of lower taxes associated with our Singapore manufacturing operation. We continue to benefit from the Australian corporate tax rate of 30% and certain Australian research and development tax benefits because we generate the majority of our taxable income in Australia.
Net Income. As a result of the factors above and share repurchases, our net income for the year ended June 30, 2012 was $254.9 million or $1.71 per diluted share compared to net income of $227.0 million or $1.44 per diluted share for the year ended June 30, 2011, an increase of 12% and 19%, respectively, over the year ended June 30, 2011.
Liquidity and Capital Resources
As of June 30, 2013 and June 30, 2012, we had cash and cash equivalents of $876.0 million and $809.5 million, respectively. Working capital was $874.8 million and $1,108.3 million at June 30, 2013 and June 30, 2012, respectively. The reduction in working capital balance is due to reclassification of our long-term debt balance of $300.0 million as a current liability in the June 2013 quarter, as the facility terminates in February 2014.
As of June 30, 2013 and June 30, 2012, our cash and cash equivalent balances held within the United States amounted to $38.2 million and $61.7 million, respectively. Our remaining cash and cash equivalent balances at June 30, 2013 and June 30, 2012, of $837.8 million and $747.8 million, respectively, were held by our non-U.S. subsidiaries, indefinitely invested outside the United States. Our cash and cash equivalent balances are held at highly rated financial institutions.
As of June 30, 2013, the cumulative amount of undistributed earnings from our foreign subsidiaries was approximately $1.6 billion, and those undistributed earnings are considered permanently reinvested. We intend to reinvest the cash and cash equivalents of those entities whose undistributed earnings are permanently reinvested in our international operations. We reassess our reinvestment assertions each reporting period and currently believe that we have sufficient sources of liquidity to support our assertion that the undistributed earnings held by foreign subsidiaries may be considered to be reinvested permanently. If these earnings had not been permanently reinvested, deferred taxes of approximately $409 million would have been recognized in our consolidated financial statements.
We repatriated $185 million and $77 million to the U.S. in fiscal years 2013 and 2012, respectively, from earnings generated in each of those years. The amount of the current year foreign earnings that we have repatriated to the U.S. in the past has been determined, and the amount that we expect to repatriate during fiscal year 2014 will be determined, based on a variety of factors, including current year earnings of our foreign subsidiaries, foreign investment needs and the cash flow needs we have in the U.S., such as for the repayment of debt, dividend distributions, and other domestic obligations. The majority of our repatriation of foreign subsidiaries earnings to the U.S. has historically occurred at year-end, although we may repatriate funds earlier in the year based on our business needs. When we repatriate funds to the U.S., we are required to pay taxes in the U.S. on these amounts based on applicable U.S. tax rates, net of any foreign tax that would be allowed to be deducted or taken as a credit against U.S. income tax. We paid $19.3 million and $2.5 million in additional U.S. federal income taxes in fiscal years 2013 and 2012, respectively, as a result of repatriation of foreign earnings generated in those years. We do not currently expect the amount of repatriated foreign earnings or the resulting additional tax expense in 2014 to differ materially from the prior fiscal year.
Inventories at June 30, 2013 decreased by $28.5 million or 16% to $145.8 million compared to June 30, 2012 inventories of $174.4 million. The decrease in inventories was due mainly to improved inventory management and the depreciation of the Australian dollar against the U.S. dollar.
- 40 -
Table of Contents
Accounts receivable, net of allowance for doubtful accounts, at June 30, 2013 were $318.3 million, an increase of $35.2 million or 12% over the June 30, 2012 accounts receivable balance of $283.2 million. The increase was slightly higher than the 11% increase in net revenues for the year ended June 30, 2013 compared to the year ended June 30, 2012, due mainly to the appreciation of the Euro against the U.S. dollar. Accounts receivable days sales outstanding of 65 days at June 30, 2013 decreased by 3 days compared to 68 days at June 30, 2012. Our allowance for doubtful accounts as a percentage of total accounts receivable at June 30, 2013 and 2012 was 3.0% and 2.5%, respectively. The credit quality of our customers remains broadly consistent with our past experience.
During the year ended June 30, 2013, we generated cash of $402.8 million from operations. This was higher than the cash generated from operations for the year ended June 30, 2012 of $383.2 million and was primarily the result of the increase in our net revenues and net income. Movements in foreign currency exchange rates during the year ended June 30, 2013 had the effect of lowering our cash and cash equivalents by $104.4 million, as reported in U.S. dollars. During fiscal years 2013 and 2012, we repurchased 4.3 million and 13.6 million shares at a cost of $188.0 million and $391.2 million, respectively. During fiscal years 2013 and 2012, we also paid dividends totaling $97.2 million and Nil, respectively.
Details of contractual obligations at June 30, 2013 are as follows:
| Payments Due by Period | ||||||||||||||||||||||||||||
| In $000s | Total | 2014 | 2015 | 2016 | 2017 | 2018 | Thereafter | |||||||||||||||||||||
| Long Term Debt | $ | 300,786 | $ | 300,017 | $ | - | $ | - | $ | - | $ | - | $ | 769 | ||||||||||||||
| Interest on Long Term Debt | 4,004 | 3,736 | 37 | 37 | 37 | 37 | 120 | |||||||||||||||||||||
| Operating Leases | 37,291 | 15,024 | 11,456 | 6,156 | 2,990 | 997 | 668 | |||||||||||||||||||||
| Purchase Obligations | 90,827 | 90,827 | - | - | - | - | - | |||||||||||||||||||||
| Total | $ | 432,908 | $ | 409,604 | $ | 11,493 | $ | 6,193 | $ | 3,027 | $ | 1,034 | $ | 1,557 | ||||||||||||||
Details of other commercial commitments at June 30, 2013 are as follows:
| Amount of Commitment Expiration Per Period | ||||||||||||||||||||||||||||
| In $000s | Total | 2014 | 2015 | 2016 | 2017 | 2018 | Thereafter | |||||||||||||||||||||
| Guarantees* |
$ | 15,307 | $ | 3,074 | $ | 715 | $ | 31 | $ | - | $ | 4,208 | $ | 7,279 | ||||||||||||||
| Other |
1,460 | 365 | 365 | 365 | 365 | - | - | |||||||||||||||||||||
| Total |
$ | 16,767 | $ | 3,439 | $ | 1,080 | $ | 396 | $ | 365 | $ | 4,208 | $ | 7,279 | ||||||||||||||
*The above guarantees mainly relate to requirements under contractual obligations with insurance companies transacting with our German subsidiaries and guarantees provided under our facility leasing obligations.
Credit Facility
During the year ended June 30, 2011, we entered into a credit agreement with lenders, including Union Bank, N.A., as Administrative Agent, Swing Line Lender and L/C Issuer, HSBC Bank USA, National Association, as Syndication Agent and Union Bank, N.A., HSBC Bank USA, National Association, Commonwealth Bank of Australia and Wells Fargo Bank, N.A. The credit agreement provides a $300 million three-year revolving credit facility, with an uncommitted option to increase the credit facility by an additional $100 million. The credit facility also includes a $10 million sublimit for letters of credit. The credit facility terminates on February 10, 2014, at which time all unpaid principal and interest under the loans must be repaid. The outstanding principal amount due under the credit facility will bear interest at a rate equal to, at our option, either (i) LIBOR plus 1.5%
- 41 -
Table of Contents
to 2.0% (depending on the applicable leverage ratio) or (ii) a base rate, as defined in the credit agreement, plus 0.5% to 1.0% (depending on the applicable leverage ratio). At June 30, 2013, the interest rate that was being charged on the outstanding principal amount was 1.7%. Commitment fees of 0.25% to 0.375% (depending on the applicable leverage ratio) apply on the unused portion of the credit facility. When we executed the credit agreement, we used a portion of the credit facilitys initial funding proceeds to repay the outstanding balance under our previously existing revolving credit facility with Union Bank, N.A., which was then terminated.
Our obligations under the credit agreement are secured by (a) the corporate stock we hold in our subsidiaries ResMed Corp. and ResMed Motor Technologies Inc. (ResMed Motor), and (b) up to 65% of the ownership interests we hold in our subsidiary ResMed EAP Holdings LLC (ResMed EAP). Our obligations under the credit agreement are also guaranteed by our subsidiaries ResMed Corp and ResMed Motor. The credit agreement contains customary covenants, including certain financial covenants and an obligation that we maintain certain financial ratios, including a maximum ratio of Funded Debt to EBITDA (each as defined in the credit agreement), an interest coverage ratio and a maximum amount of annual capital expenditures. The entire principal amount of the credit facility and any accrued but unpaid interest may be declared immediately due and payable if an event of default occurs. Events of default include failure to make payments when due, a default in the performance of any covenants in the credit agreement or related documents or certain changes of control of us or our subsidiaries ResMed Corp., ResMed Motor, ResMed Limited, ResMed Holdings Ltd/LLC or ResMed EAP.
On January 25, 2012, we entered into a first amendment to the credit agreement. The amendment increased, from $300 million to $400 million, the maximum principal amount that can be borrowed on a revolving basis under the credit agreement, subject to customary conditions
At June 30, 2013, we were in compliance with our debt covenants and there was $300.0 million outstanding under the credit agreement. As the credit facility terminates on February 10, 2014, we have begun negotiating a new financing facility. We expect that the new facility will have similar terms to the existing facility and that it will be in place before our existing facility expires.
We expect to satisfy all of our liquidity requirements through a combination of cash on hand, cash generated from operations and debt facilities.
Tax Expense
Our income tax rate is governed by the laws of the regions in which our income is recognized. To date, a substantial portion of our income has been subject to income tax in Australia where the statutory rate was 30% in fiscal years 2013, 2012 and 2011. During fiscal years 2013, 2012 and 2011, our consolidated effective tax rate has fluctuated between approximately 20% and approximately 25%. These fluctuations have resulted from, and future effective tax rates will depend on, numerous factors, including the amount of research and development expenditures for which an additional Australian tax credit is available, the geographic mix of taxable income and other tax credits or benefits available to us under applicable tax laws including the lower statutory tax rates and other incentives associated with our Singapore and Malaysia manufacturing operations.
We account for income taxes under the asset and liability method. Deferred tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that includes the enactment date.
- 42 -
Table of Contents
Critical Accounting Principles and Estimates
The preparation of financial statements in conformity with accounting principles generally accepted in the United States requires us to make estimates and judgments that affect our reported amounts of assets and liabilities, revenues and expenses and related disclosures of contingent assets and liabilities. We evaluate our estimates on an ongoing basis, including those estimates related to allowance for doubtful accounts, inventory adjustments, warranty obligations, goodwill, impaired assets, intangible assets, income taxes, deferred tax valuation allowances and stock-based compensation costs.
We state these accounting policies in the Notes to the consolidated financial statements and at relevant sections in this discussion and analysis. The estimates are based on the information that is currently available to us and on various other assumptions that we believe to be reasonable under the circumstances. Actual results could vary from those estimates under different assumptions or conditions.
We believe that the following critical accounting policies affect the more significant judgments and estimates used in the preparation of our consolidated financial statements:
(1) Allowance for Doubtful Accounts. We maintain an allowance for doubtful accounts for estimated losses resulting from the inability of our customers to make required payments, which results in bad debt expense. We determine the adequacy of this allowance by periodically evaluating individual customer receivables, considering a customers financial condition, credit history and current economic conditions. If the financial condition of our customers were to deteriorate, resulting in an impairment of their ability to make payments, additional allowances may be required.
(2) Inventory Adjustments. Inventories are stated at lower of cost or market and are determined by the first-in, first-out method. We review the components of inventory on a regular basis for excess, obsolete and impaired inventory based on estimated future usage and sales. The likelihood of any material inventory write-downs depends on changes in competitive conditions, new product introductions by us or our competitors, or rapid changes in customer demand.
(3) Valuation of Goodwill, Intangible and Other Long-Lived Assets. We make assumptions in establishing the carrying value, fair value and estimated lives of our goodwill, intangibles and other long-lived assets. The criteria used for these evaluations include managements estimate of the assets continuing ability to generate positive income from operations and positive cash flow in future periods compared to the carrying value of the asset, as well as the strategic significance of any identifiable intangible asset in our business objectives. If assets are considered to be impaired, we recognize as impairment the amount by which the carrying value of the assets exceeds their fair value. We base useful lives and related amortization or depreciation expense on our estimate of the period that the assets will generate revenues or otherwise be used by us. Factors that would influence the likelihood of a material change in our reported results include significant changes in the assets ability to generate positive cash flow, loss of legal ownership or title to the asset, a significant decline in the economic and competitive environment on which the asset depends, significant changes in our strategic business objectives, utilization of the asset, and a significant change in the economic and/or political conditions in certain countries.
We conducted our annual review for goodwill impairment during the final quarter of fiscal 2013 using a quantitative assessment. The results of our annual review indicated that no impaired goodwill exists as the fair value for each reporting unit significantly exceeded its carrying value.
(4) Income Tax. We assess our income tax positions and record tax benefits for all years subject to examination based upon managements evaluation of the facts, circumstances, and information
- 43 -
Table of Contents
available at the reporting date. Where we determine that it is not more likely than not that we would be able to realize all or part of our net deferred tax assets in the future, an adjustment to the deferred tax assets would be charged to income tax expense in the period such determination is made. Likewise, if we later determine that it is more likely than not that the net deferred tax assets would be realized, the previously provided valuation allowance would be reversed. These changes to the valuation allowance, and resulting increases or decreases in income tax expense, could have a material effect on our operating results.
Our income tax returns are based on calculations and assumptions that are subject to examination by various tax authorities. In addition, the calculation of our tax liabilities involves dealing with uncertainties in the application of complex tax laws. We recognize liabilities for uncertain tax positions based on a two-step process. The first step is to evaluate the tax position for recognition by determining if the weight of available evidence indicates that it is more likely than not that the position will be sustained on audit, including resolution of related appeals or litigation processes, if any. The second step is to measure the tax benefit as the largest amount that is more than 50% likely of being realized upon settlement. While we believe we have appropriate support for the positions taken on our tax returns, we regularly assess the potential outcomes of examinations by tax authorities in determining the adequacy of our provision for income taxes, and adjust the income tax provision, income taxes payable and deferred taxes in the period in which the facts that give rise to a revision become known.
(5) Provision for Warranty. We provide for the estimated cost of product warranties at the time the related revenue is recognized. We determine the amount of this provision by using a financial model, which takes into consideration actual historical expenses and potential risks associated with our different products. We use this financial model to calculate the future probable expenses related to warranty and the required level of the warranty provision. Although we engage in product improvement programs and processes, our warranty obligation is affected by product failure rates and costs incurred to correct those product failures. Should actual product failure rates or estimated costs to repair those product failures differ from our estimates, we would be required to revise our estimated warranty provision.
(6) Revenue Recognition. We generally record revenue on product sales at the time of shipment, which is when title transfers to the customer. We do not record revenue on product sales that require customer acceptance until we receive acceptance. We initially defer service revenue received in advance from service contracts and recognize that deferred revenue ratably over the life of the service contract. We initially defer revenue we receive in advance from rental unit contracts and recognize that deferred revenue ratably over the life of the rental contract. Otherwise, we recognize revenue from rental unit contracts ratably over the life of the rental contract. We include in revenue freight charges we bill to customers. We charge all freight-related expenses to cost of sales. Taxes assessed by government authorities that are imposed on and concurrent with revenue-producing transactions, such as sales and value added taxes, are excluded from revenue.
We do not normally offer a right of return or other recourse with respect to the sale of our products, other than returns for product defects or other warranty claims. We do not recognize revenues if we offer a right of return or variable sale prices for subsequent events or activities. However, as part of our sales processes we may provide upfront discounts for large orders, one-time special pricing to support new product introductions, sales rebates for centralized purchasing entities or price-breaks for regular order volumes. We record the costs of all such programs as an adjustment to revenue. Our products are predominantly therapy-based equipment and require no installation. Therefore, we have no significant installation obligations.
(7) Stock-Based Compensation. We measure the compensation cost of all stock-based awards at fair value on the date of grant. We recognize that value as compensation expense over the service period,
- 44 -
Table of Contents
net of estimated forfeitures. We estimate the fair value of employee stock options and purchase rights granted under our Employee Stock Purchase Plan using a Black-Scholes valuation model. The fair value of an award is affected by our stock price on the date of grant as well as other assumptions including the estimated volatility of our stock price over the term of the awards, the expected dividend per share and the expected life of the awards. The risk-free interest rate assumption we use is based upon the U.S. Treasury yield curve at the time of grant appropriate for the expected life of the awards. Expected volatilities are based on a combination of historical volatilities of our stock and the implied volatilities from tradeable options of our stock corresponding to the expected term of the options. We use a combination of the historic and implied volatilities as the addition of the implied volatility is more representative of our future stock price trends. While there is a tradeable market of options on our common stock, less emphasis is placed on the implied volatility of these options due to the relative low volumes of these traded options and the difference in the terms compared to our employee options. In order to determine the estimated period of time that we expect employees to hold their stock options, we use historical rates by employee groups. The estimation of stock awards that will ultimately vest requires judgment, and to the extent actual results differ from our estimates, such amounts will be recorded as a cumulative adjustment in the period estimates are revised. The aforementioned inputs entered into the Black-Scholes valuation model we use to fair value our stock awards are subjective estimates and changes to these estimates will cause the fair value of our stock awards and related stock-based compensation expense we record to vary.
We estimate the fair value of restricted stock units based on the market value of the underlying shares as determined at the grant date less the fair value of dividends that holders are not entitled to, during the vesting period. We estimate the weighted average grant date fair value of performance restricted stock units (PRSUs), which contain a market condition, using a Monte-Carlo simulation valuation model.
Recently Issued Accounting Pronouncements
See Note 3 New Accounting Pronouncements to the consolidated financial statements for a description of recently issued accounting pronouncements, including the expected dates of adoption and estimated effects on our results of operations, financial positions and cash flows.
Off-Balance Sheet Arrangements
As of June 30, 2013, we are not involved in any significant off-balance sheet arrangements, as defined in Item 303(a)(4)(ii) of Regulation S-K promulgated by the SEC.
| ITEM 7A | QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET AND BUSINESS RISKS |
Foreign Currency Market Risk
Our reporting currency is the U.S. dollar, although the financial statements of our non-U.S. subsidiaries are maintained in their respective local currencies. We transact business in various foreign currencies, including a number of major European currencies as well as the Australian dollar. We have significant foreign currency exposure through both our Australian and Singapore manufacturing activities and international sales operations. We have established a foreign currency hedging program using purchased currency options and forward contracts to hedge foreign-currency-denominated financial assets, liabilities and manufacturing cash flows. The goal of this hedging program is to economically manage the financial impact of foreign currency exposures predominantly denominated in euros, Australian dollars and Singapore dollars. Under this program, increases or decreases in our foreign-currency-denominated financial assets, liabilities, and firm commitments are partially offset by gains and losses on the hedging instruments. We do not enter into financial instruments for trading or speculative purposes. The foreign currency derivatives portfolio is recorded
- 45 -
Table of Contents
in the consolidated balance sheets at fair value and included in other assets or other liabilities. All movements in the fair value of the foreign currency derivatives are recorded within other income, net, on our consolidated statements of income.
The table below provides information (in U.S. dollars) on our foreign-currency-denominated financial assets by legal entity functional currency as of June 30, 2013 (in thousands):
| Australian Dollar (AUD) |
U.S. Dollar (USD) |
Euro (EUR) |
Singapore Dollar (SGD) |
Canadian Dollar (CAD) |
Japanese Yen (JPY) |
Malaysian Ringgit (MYR) |
||||||||||||||||||||||
| AUD Functional: |
||||||||||||||||||||||||||||
| Assets |
- | 113,773 | 67,219 | 1,199 | - | 10 | 3,850 | |||||||||||||||||||||
| Liability |
- | (59,124 | ) | (21,942 | ) | (5 | ) | - | (1,409 | ) | - | |||||||||||||||||
| Foreign Currency Hedges |
- | (35,000 | ) | (52,035 | ) | - | - | - | - | |||||||||||||||||||
| Net Total |
- | 19,649 | (6,758 | ) | 1,194 | - | (1,399 | ) | 3,850 | |||||||||||||||||||
| USD Functional: |
||||||||||||||||||||||||||||
| Assets |
- | - | - | - | 10,146 | - | - | |||||||||||||||||||||
| Liability |
- | - | (300 | ) | - | - | - | - | ||||||||||||||||||||
| Foreign Currency Hedges |
- | - | - | - | (8,076 | ) | - | - | ||||||||||||||||||||
| Net Total |
- | - | (300 | ) | - | 2,070 | - | - | ||||||||||||||||||||
| EURO Functional: |
||||||||||||||||||||||||||||
| Assets |
14 | 359 | - | - | - | - | - | |||||||||||||||||||||
| Liability |
(7 | ) | (2,075 | ) | - | - | - | - | - | |||||||||||||||||||
| Foreign Currency Hedges |
- | - | - | - | - | - | - | |||||||||||||||||||||
| Net Total |
7 | (1,716 | ) | - | - | - | - | - | ||||||||||||||||||||
| GBP Functional: |
||||||||||||||||||||||||||||
| Assets |
- | 32 | 11,502 | - | - | - | - | |||||||||||||||||||||
| Liability |
- | - | (10,130 | ) | - | - | - | - | ||||||||||||||||||||
| Foreign Currency Hedges |
- | - | - | - | - | - | - | |||||||||||||||||||||
| Net Total |
- | 32 | 1,372 | - | - | - | - | |||||||||||||||||||||
| SGD Functional : |
||||||||||||||||||||||||||||
| Assets |
297 | 34,500 | 59,650 | - | - | - | - | |||||||||||||||||||||
| Liability |
(2,182 | ) | (84,447 | ) | (27,962 | ) | - | - | - | - | ||||||||||||||||||
| Foreign Currency Hedges |
2,280 | 40,000 | (39,026 | ) | - | - | - | - | ||||||||||||||||||||
| Net Total |
395 | (9,947 | ) | (7,338 | ) | - | - | - | - | |||||||||||||||||||
| INR Functional : |
||||||||||||||||||||||||||||
| Assets |
- | 238 | 1 | - | - | - | - | |||||||||||||||||||||
| Liability |
- | (1,649 | ) | (404 | ) | - | - | - | - | |||||||||||||||||||
| Foreign Currency Hedges |
- | - | - | - | - | - | - | |||||||||||||||||||||
| Net Total |
- | (1,411 | ) | (403 | ) | - | - | - | - | |||||||||||||||||||
| MYR Functional: |
||||||||||||||||||||||||||||
| Assets |
- | 3,456 | 72 | - | - | - | - | |||||||||||||||||||||
| Liability |
(35 | ) | (3,389 | ) | - | (17 | ) | - | - | - | ||||||||||||||||||
| Foreign Currency Hedges |
- | - | - | - | - | - | - | |||||||||||||||||||||
| Net Total |
(35 | ) | 67 | 72 | (17 | ) | - | - | - | |||||||||||||||||||
- 46 -
Table of Contents
The table below provides information about our foreign currency derivative financial instruments and presents the information in U.S. dollar equivalents. The table summarizes information on instruments and transactions that are sensitive to foreign currency exchange rates, including foreign currency call options, collars and forward contracts held at June 30, 2013. The table presents the notional amounts and weighted average exchange rates by contractual maturity dates for our foreign currency derivative financial instruments. These notional amounts generally are used to calculate payments to be exchanged under the options contracts (in thousands, except exchange rates):
|
Foreign Exchange |
Fair Value Assets / (Liabilities) |
|||||||||||
| FY 2014 | FY 2015 | FY 2016 | Total | June 30, 2013 |
June 30, 2012 |
|||||||
| Receive AUD/Pay USD |
||||||||||||
| Contract amount |
70,000 | 10,000 | - | 80,000 | (822) | 4,171 | ||||||
| Ave. contractual |
AUD 1 = USD 0.9952 | AUD 1 = USD 1.0500 | AUD 1 = USD 1.0017 | |||||||||
| Receive AUD/Pay Euro |
||||||||||||
| Contract amount |
176,000 | 117,000 | - | 293,000 | (6,985) | 10,592 | ||||||
| Ave. contractual |
AUD 1 = Euro 0.7697 | AUD 1 = Euro 0.8120 | AUD 1 = Euro 0. 7861 | |||||||||
| Receive SGD/Pay Euro |
||||||||||||
| Contract amount |
39,000 | - | - | 39,000 | 501 | (145) | ||||||
| Ave. contractual |
SGD 1 = Euro 0. 5982 | SGD 1 = Euro 0. 5982 | ||||||||||
| Receive AUD/Pay SGD |
||||||||||||
| Contract amount |
2,000 | - | - | 2,000 | (193) | 16 | ||||||
| Ave. contractual |
SGD 1 = AUD 0.7968 | SGD 1 = AUD 0.7968 | ||||||||||
| Receive USD/Pay SGD |
||||||||||||
| Contract amount |
40,000 | - | - | 40,000 | 284 | - | ||||||
| Ave. contractual |
SGD 1 = USD 0.7938 | SGD 1 = USD 0.7938 | ||||||||||
| Receive CHF/Pay AUD |
||||||||||||
| Contract amount |
- | - | - | - | - | (3) | ||||||
| Ave. contractual |
||||||||||||
| Receive USD/Pay CAD |
||||||||||||
| Contract amount |
8,000 | - | - | 8,000 | 215 | - | ||||||
| Ave. contractual |
USD 1 = CAD 1.0251 | CAD 1 = USD 1.0251 | ||||||||||
Interest Rate Risk
We are exposed to risk associated with changes in interest rates affecting the return on our cash and cash equivalents and debt. At June 30, 2013, we maintained cash and cash equivalents of $876.0 million principally comprised of bank term deposits and at call accounts and are invested at both short-term fixed interest rates and variable interest rates. At June 30, 2013, we had total long-term debt, including the current portion of those obligations, of $300.8 million of which, $300.0 million is subject to variable interest rates. A hypothetical 10% change in interest rates during the year ended June 30, 2013, would not have had a material impact on pretax income. We have no interest rate hedging agreements.
- 47 -
Table of Contents
| ITEM 8 | CONSOLIDATED FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA |
The information required by this Item is incorporated by reference to the financial statements set forth in Item 15 of Part IV of this report, Exhibits and Consolidated Financial Statement Schedules.
| a) | Index to Consolidated Financial Statements |
| Report of Independent Registered Public Accounting Firm |
F1 | |||
| Consolidated Balance Sheets as of June 30, 2013 and 2012 |
F2 | |||
| Consolidated Statements of Income for the years ended June 30, 2013, 2012 and 2011 |
F3 | |||
| Consolidated Statements of Comprehensive Income for the years ended June 30, 2013, 2012 and 2011 |
F4 | |||
| Consolidated Statements of Stockholders Equity for the years ended June 30, 2013, 2012 and 2011 |
F5 | |||
| Consolidated Statements of Cash Flows for the years ended June 30, 2013, 2012 and 2011 |
F6 | |||
| Notes to Consolidated Financial Statements |
F7 | |||
| Schedule II Valuation and Qualifying Accounts and Reserves |
| b) | Supplementary Data |
Quarterly Financial Information (unaudited) The quarterly results for the years ended June 30, 2013 and 2012 are summarized below (in thousands, except per share amounts):
| 2013 | First Quarter |
Second Quarter |
Third Quarter |
Fourth Quarter |
Fiscal Year | |||||||||||||||
| Net revenues |
$ | 339,731 | $ | 376,538 | $ | 383,581 | $ | 414,607 | $ | 1,514,457 | ||||||||||
| Gross profit |
208,648 | 232,712 | 239,449 | 259,848 | 940,657 | |||||||||||||||
| Net income |
71,265 | 77,942 | 84,913 | 73,013 | 307,133 | |||||||||||||||
| Basic earnings per share |
0.50 | 0.54 | 0.59 | 0.51 | 2.15 | |||||||||||||||
| Diluted earnings per share |
0.49 | 0.53 | 0.58 | 0.50 | 2.10 | |||||||||||||||
| 2012 | First Quarter |
Second Quarter |
Third Quarter |
Fourth Quarter |
Fiscal Year | |||||||||||||||
| Net revenues |
$ | 314,775 | $ | 332,738 | $ | 349,073 | $ | 371,929 | $ | 1,368,515 | ||||||||||
| Gross profit |
185,055 | 198,715 | 210,496 | 226,469 | 820,735 | |||||||||||||||
| Net income |
50,518 | 62,872 | 64,613 | 76,847 | 254,850 | |||||||||||||||
| Basic earnings per share |
0.33 | 0.43 | 0.45 | 0.54 | 1.75 | |||||||||||||||
| Diluted earnings per share |
0.32 | 0.42 | 0.44 | 0.53 | 1.71 | |||||||||||||||
Note: Per share amounts for each quarter are computed independently, and, due to the computation formula, the sum of the four quarters may not equal the year.
| ITEM 9 | CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE |
None.
| ITEM 9A | CONTROLS AND PROCEDURES |
We maintain disclosure controls and procedures that are designed to ensure that information required to be disclosed in our Exchange Act reports is recorded, processed, summarized and reported within the time periods specified in the Securities and Exchange Commissions rules and forms and that such information is accumulated and communicated to our management, including our chief executive officer and chief financial officer, as appropriate, to allow for timely decisions regarding required disclosure. In designing and evaluating the disclosure controls and procedures, management
- 48 -
Table of Contents
recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired control objectives, and management is required to apply its judgment in evaluating the cost-benefit relationship of possible controls and procedures.
As required by SEC Rule 13a-15(b), we carried out an evaluation, under the supervision and with the participation of our management, including our chief executive officer and chief financial officer, of the effectiveness of the design and operation of our disclosure controls and procedures as of June 30, 2013. Based on the foregoing, our chief executive officer and chief financial officer concluded that our disclosure controls and procedures were effective at the reasonable assurance level as of June 30, 2013.
There has been no change in our internal controls over financial reporting during our most recent fiscal quarter that has materially affected, or is reasonably likely to materially affect, our internal controls over financial reporting.
- 49 -
Table of Contents
MANAGEMENTS REPORT ON INTERNAL CONTROL OVER FINANCIAL REPORTING
Our management is responsible for establishing and maintaining adequate internal control over financial reporting as defined in Rules 13a-15(f) and 15d-15(f) under the Securities Exchange Act of 1934. Our internal control over financial reporting is designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles in the United States of America. Our internal control over financial reporting includes those policies and procedures that:
| (i) | Pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of our assets; |
| (ii) | Provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that our receipts and expenditures are being made only in accordance with authorizations of our management and directors; and |
| (iii) | Provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of our assets that could have a material effect on the financial statements. |
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Management assessed the effectiveness of our internal control over financial reporting as of June 30, 2013. Management based this assessment on criteria for effective internal control over financial reporting described in Internal Control Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commissions 1992 framework. Managements assessment included an evaluation of the design of our internal control over financial reporting and testing of the operational effectiveness of its internal control over financial reporting. Management reviewed the results of its assessment with the audit committee of our board of directors.
Based on our assessment and those criteria, management has concluded that we maintained effective internal control over financial reporting as of June 30, 2013.
KPMG LLP, independent registered public accounting firm, who audited and reported on the consolidated financial statements of ResMed, Inc. included in this report, has issued an attestation report on the effectiveness of internal control over financial reporting.
- 50 -
Table of Contents
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
The Board of Directors and Stockholders
ResMed Inc.:
We have audited the internal control over financial reporting of ResMed Inc. as of June 30, 2013, based on criteria established in Internal Control Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO). The management of ResMed Inc. is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting, included in the accompanying Managements Report on Internal Control Over Financial Reporting. Our responsibility is to express an opinion on the internal control over financial reporting of ResMed Inc. based on our audit.
We conducted our audit in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects. Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, and testing and evaluating the design and operating effectiveness of internal control based on the assessed risk. Our audit also included performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
A companys internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A companys internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the companys assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
In our opinion, ResMed Inc. maintained, in all material respects, effective internal control over financial reporting as of June 30, 2013, based on criteria established in Internal Control Integrated Framework issued by COSO.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), the consolidated balance sheets of ResMed Inc. and subsidiaries as of June 30, 2013 and 2012, and the related consolidated statements of income, comprehensive income, stockholders equity, and cash flows for each of the years in the three-year period ended June 30, 2013, and the related financial statement schedule, and our report dated August 12, 2013 expressed an unqualified opinion on those consolidated financial statements and financial statement schedule.
| /s/ KPMG LLP |
| San Diego, California |
| August 12, 2013 |
| ITEM 9B | OTHER INFORMATION |
None.
- 51 -
Table of Contents
PART III
| ITEM 10 | DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE |
Information required by this Item is incorporated by reference from our definitive proxy statement for our November 14, 2013, annual meeting of stockholders, which will be filed with the Securities and Exchange Commission within 120 days after June 30, 2013.
We have filed as exhibits to this annual report on Form 10-K for the year ended June 30, 2013, the certifications of our chief executive officer and chief financial officer required by Section 302 of the Sarbanes-Oxley Act of 2002.
| ITEM 11 | EXECUTIVE COMPENSATION |
Information required by this Item is incorporated by reference from our definitive proxy statement for our November 14, 2013, annual meeting of stockholders, which will be filed with the Securities and Exchange Commission within 120 days after June 30, 2013.
| ITEM 12 | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS |
Information required by this Item is incorporated by reference from our definitive proxy statement for our November 14, 2013, annual meeting of stockholders, which will be filed with the Securities and Exchange Commission within 120 days after June 30, 2013.
| ITEM 13 | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE |
Information required by this Item is incorporated by reference from our definitive proxy statement for our November 14, 2013, annual meeting of stockholders, which will be filed with the Securities and Exchange Commission within 120 days after June 30, 2013.
| ITEM 14 | PRINCIPAL ACCOUNTING FEES AND SERVICES |
Information required by this Item is incorporated by reference from our definitive proxy statement for our November 14, 2013, annual meeting of stockholders, which will be filed with the Securities and Exchange Commission within 120 days after June 30, 2013.
- 52 -
Table of Contents
PART IV
| ITEM 15 | EXHIBITS AND CONSOLIDATED FINANCIAL STATEMENT SCHEDULES |
The following documents are filed as part of this report:
| (a) | Consolidated Financial Statements and Schedules The index to our consolidated financial statements and schedules are set forth in the Index to Consolidated Financial Statements under Item 8 of this report. | |
| (b) | Exhibit Lists | |
| 3.1 | First Restated Certificate of Incorporation of Registrant, as amended. (5) | |
| 3.2 | Fifth Amended and Restated Bylaws of ResMed Inc. (17) | |
| 4.1 | Form of certificate evidencing shares of Common Stock. (1) | |
| 10.1 | Licensing Agreement between the University of Sydney and ResMed Ltd dated May 17, 1991, as amended. (1) | |
| 10.2* | ResMed Inc. 2006 Incentive Award Plan. (6) | |
| 10.3* | Amendment No. 1 to the ResMed Inc. 2006 Incentive Award Plan. (3) | |
| 10.4* | 2006 Grant agreement for Board of Directors. (3) | |
| 10.5* | 2006 Grant agreement for Executive Officers. (5) | |
| 10.6* | 2006 Grant agreement for Australian Executive Officers. (5) | |
| 10.7* | Form of Executive Agreement. (4) | |
| 10.8* | Amended and Restated 2006 Incentive Award Plan dated November 20, 2008. (7) | |
| 10.9 | Departure of Directors or Certain Officers dated December 12, 2008. (8) | |
| 10.10 | Approval of new share repurchase program dated May 29, 2009. (9) | |
| 10.11 | Form of Indemnification Agreements for our directors and officers. (10) | |
| 10.12 | Form of Access Agreement for directors. (10) | |
| 10.13* | Updated Form of Executive Agreement. (2)(16) | |
| 10.14 | ResMed Inc. 2009 Incentive Award Plan. (11) | |
| 10.15 | ResMed Inc. 2009 Employee Stock Purchase Plan. (11) | |
| 10.16 | Amendment No. 1 to the ResMed Inc. 2009 Employee Stock Purchase Plan (18) | |
| 10.17 | Form of Restricted Stock Award Agreement. (11) | |
| 10.18 | ResMed Inc. Deferred Compensation Plan. (12) | |
| 10.19 | Credit Agreement, dated February 10, 2011, by and between ResMed Inc. and the lenders, including Union Bank, N.A., HSBC Bank USA, National Association, Commonwealth Bank of Australia and Wells Fargo Bank, N.A. (13) | |
| 10.20 | First Amendment to Credit Agreement, dated January 25, 2012, by and between ResMed Inc. and the lenders, including Union Bank, N.A., HSBC Bank USA, National Association, Commonwealth Bank of Australia and Wells Fargo Bank, N.A. (15) | |
- 53 -
Table of Contents
| 10.21 | Pledge and Security Agreement, dated as of February 10, 2011, by and between ResMed Inc., as Pledgor, and Union Bank, N.A., as Administrative Agent. (13) | |
| 10.22 | Unconditional Guaranty entered into as of February 10, 2011, by each of ResMed Corp., ResMed Assembly US Inc. and ResMed Motor Technologies Inc., in favor of Union Bank, N.A., as Administrative Agent. (13) | |
| 10.23 | Form of Restricted Stock Unit Award Agreement for Executive Officers. (14) | |
| 10.24 | Form of Restricted Stock Unit Award Agreement for Directors. (14) | |
| 10.25 | Form of Stock Option Grant for Executive Officers. (14) | |
| 10.26 | Form of Stock Option Grant for Directors. (14) | |
| 10.27 | Form of Performance-Based Restricted Stock Unit Award Agreement for Executive Officers. (19) | |
| 21.1 | Subsidiaries of the Registrant. (20) | |
| 23.1 | Consent of Independent Registered Public Accounting Firm. (20) | |
| 31.1 | Certification of Chief Executive Officer Pursuant to Section 302 of Sarbanes-Oxley Act of 2002. (20) | |
| 31.2 | Certification of Chief Financial Officer Pursuant to Section 302 of Sarbanes-Oxley Act of 2002. (20) | |
| 32.1 | Certification of Chief Executive Officer and Chief Financial Officer pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. (20) | |
| 101 | The following materials from ResMed Inc.s Annual Report on Form 10-K for the fiscal year ended June 30, 2013 formatted in Extensible Business Reporting Language (XBRL): (i) the Consolidated Balance Sheets, (ii) the Consolidated Statements of Income, (iii) the Consolidated Statements of Stockholders Equity and Comprehensive Income, (iv) the Consolidated and Statements of Cash Flows and (v) related notes.
|
|
* Management contract or compensatory plan or arrangement
(1) Incorporated by reference to the Registrants Registration Statement on Form S-1 (No. 33-91094) declared effective on June 1, 1995.
(2) Incorporated by reference to the Registrants Report on Form 10-K for the year ended June 30, 2009.
(3) Incorporated by reference to the Registrants Report on Form 10-Q for the quarter ended December 31, 2006.
(4) Incorporated by reference to the Registrants Report on Form 8-K filed on July 13, 2007.
(5) Incorporated by reference to the Registrants Report on Form 10-K for the year ended June 30, 2007.
(6) Incorporated by reference to the Registrants Report on Form 8-K filed on November 15, 2006.
(7) Incorporated by reference to Appendix A of the Registrants Definitive Proxy Statement filed on October 15, 2008.
(8) Incorporated by reference to the Registrants Report on Form 8-K filed on December 15, 2008.
(9) Incorporated by reference to the Registrants Report on Form 8-K filed on June 4, 2009.
(10) Incorporated by reference to the Registrants Report on Form 8-K filed on June 24, 2009.
(11) Incorporated by reference to the Registrants Report on Form 8-K filed on November 23, 2009.
(12) Incorporated by reference to the Registrants Report on Form 8-K filed on May 25, 2010.
(13) Incorporated by reference to the Registrants Report on Form 8-K filed on February 14, 2011.
(14) Incorporated by reference to the Registrants Report on Form 10-Q for the quarter ended September 30, 2011.
(15) Incorporated by reference to the Registrants Report on Form 8-K filed on January 26, 2012.
(16) Incorporated by reference to the Registrants Report on Form 8-K filed on July 2, 2012.
(17) Incorporated by reference to the Registrants Report on Form 8-K/A filed on September 17, 2012.
(18) Incorporated by reference to Appendix A of the Registrants Definitive Proxy Statement filed on October 4, 2012.
(19) Incorporated by reference to the Registrants Report on Form 8-K filed on November 21, 2012.
(20) Filed with this report.
- 54 -
Table of Contents
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
The Board of Directors and Stockholders
ResMed Inc.:
We have audited the accompanying consolidated balance sheets of ResMed Inc. and subsidiaries (the Company) as of June 30, 2013 and 2012, and the related consolidated statements of income, comprehensive income, stockholders equity, and cash flows for each of the years in the three-year period ended June 30, 2013. In connection with our audits of the consolidated financial statements, we also have audited financial statement schedule II. These consolidated financial statements and financial statement schedule are the responsibility of the Companys management. Our responsibility is to express an opinion on these consolidated financial statements and financial statement schedule based on our audits.
We conducted our audits in accordance with the standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our opinion.
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of ResMed Inc. and subsidiaries as of June 30, 2013 and 2012, and the results of their operations and their cash flows for each of the years in the three-year period ended June 30, 2013, in conformity with U.S. generally accepted accounting principles. Also in our opinion, the related financial statement schedule, when considered in relation to the basic consolidated financial statements taken as a whole, presents fairly, in all material respects, the information set forth therein.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States), the Companys internal control over financial reporting as of June 30, 2013, based on criteria established in Internal ControlIntegrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO), and our report dated August 12, 2013, expressed an unqualified opinion on the effectiveness of the internal control over financial reporting of ResMed Inc.
/s/ KPMG LLP
San Diego, California
August 12, 2013
- F1 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Consolidated Balance Sheets
June 30, 2013 and 2012
(In thousands, except share and per share data)
|
June 30, 2013 |
June 30, 2012 |
|||||||
| Assets |
||||||||
| Current assets: |
||||||||
| Cash and cash equivalents |
$ | 876,048 | $ | 809,541 | ||||
| Accounts receivable, net of allowance for doubtful accounts of $9,912 and $7,313 at June 30, 2013 and June 30, 2012, respectively |
318,349 | 283,160 | ||||||
| Inventories (note 5) |
145,847 | 174,351 | ||||||
| Deferred income taxes (note 14) |
38,552 | 19,590 | ||||||
| Income taxes receivable |
8,910 | 2,282 | ||||||
| Prepaid expenses and other current assets |
61,143 | 72,227 | ||||||
| Total current assets |
1,448,849 | 1,361,151 | ||||||
| Non-current assets: |
||||||||
| Property, plant and equipment, net (note 6) |
411,433 | 434,363 | ||||||
| Goodwill and other intangible assets, net (note 7) |
324,468 | 311,036 | ||||||
| Deferred income taxes (note 14) |
20,053 | 23,500 | ||||||
| Other assets |
5,918 | 7,819 | ||||||
| Total non-current assets |
761,872 | 776,718 | ||||||
| Total assets |
$ | 2,210,721 | $ | 2,137,869 | ||||
| Liabilities and Stockholders Equity |
||||||||
| Current liabilities: |
||||||||
| Accounts payable |
$ | 60,688 | $ | 55,006 | ||||
| Accrued expenses (note 9) |
137,674 | 127,381 | ||||||
| Deferred revenue |
44,953 | 41,563 | ||||||
| Income taxes payable |
30,090 | 27,777 | ||||||
| Deferred income taxes (note 14) |
627 | 1,073 | ||||||
| Current portion of long-term debt (note 11) |
300,017 | 52 | ||||||
| Total current liabilities |
574,049 | 252,852 | ||||||
| Non-current liabilities: |
||||||||
| Deferred income taxes (note 14) |
9,895 | 8,843 | ||||||
| Deferred revenue |
11,928 | 14,384 | ||||||
| Long-term debt (note 11) |
769 | 250,783 | ||||||
| Income taxes payable |
3,564 | 3,380 | ||||||
| Total non-current liabilities |
26,156 | 277,390 | ||||||
| Total liabilities |
600,205 | 530,242 | ||||||
| Commitments and contingencies (notes 18 and 19) |
||||||||
| Stockholders equity: (note 12) |
||||||||
| Preferred stock, $0.01 par value, 2,000,000 shares authorized; none issued |
- | - | ||||||
| Common stock, $0.004 par value, 350,000,000 shares authorized; 174,038,766 issued and 142,013,753 outstanding at June 30, 2013 and 169,752,781 issued and 142,021,032 outstanding at June 30, 2012 |
568 | 568 | ||||||
| Additional paid-in capital |
1,025,064 | 899,717 | ||||||
| Retained earnings |
1,576,641 | 1,366,712 | ||||||
| Treasury stock, at cost, 32,026,013 shares at June 30, 2013, and 27,731,749 shares at June 30, 2012 |
(1,083,845 | ) | (895,826 | ) | ||||
| Accumulated other comprehensive income |
92,088 | 236,456 | ||||||
| Total stockholders equity |
1,610,516 | 1,607,627 | ||||||
| Total liabilities and stockholders equity |
$ | 2,210,721 | $ | 2,137,869 | ||||
See accompanying notes to consolidated financial statements.
- F2 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Consolidated Statements of Income
Years Ended June 30, 2013, 2012 and 2011
(In thousands, except per share data)
|
June 30, 2013 |
June 30, 2012 |
June 30, 2011 |
||||||||||
| Net revenue |
$ | 1,514,457 | $ | 1,368,515 | $ | 1,243,148 | ||||||
| Cost of sales |
573,800 | 547,780 | 501,822 | |||||||||
| Gross profit |
940,657 | 820,735 | 741,326 | |||||||||
| Operating expenses: |
||||||||||||
| Selling, general and administrative |
430,802 | 402,621 | 372,249 | |||||||||
| Research and development |
120,124 | 109,733 | 92,007 | |||||||||
| Education, research and settlement charge (note 23) |
24,765 | - | - | |||||||||
| Amortization of acquired intangible assets |
10,142 | 13,974 | 10,146 | |||||||||
| Total operating expenses |
585,833 | 526,328 | 474,402 | |||||||||
| Income from operations |
354,824 | 294,407 | 266,924 | |||||||||
| Other income, net: |
||||||||||||
| Interest income |
38,873 | 33,866 | 27,801 | |||||||||
| Interest expense |
(6,387 | ) | (4,786 | ) | (1,758 | ) | ||||||
| Other, net (note 13) |
(2,191 | ) | 8,458 | 10,740 | ||||||||
| Total other income, net |
30,295 | 37,538 | 36,783 | |||||||||
| Income before income taxes |
385,119 | 331,945 | 303,707 | |||||||||
| Income taxes (note 14) |
77,986 | 77,095 | 76,721 | |||||||||
| Net income |
$ | 307,133 | $ | 254,850 | $ | 226,986 | ||||||
| Basic earnings per share |
$ | 2.15 | $ | 1.75 | $ | 1.49 | ||||||
| Diluted earnings per share (note 4) |
$ | 2.10 | $ | 1.71 | $ | 1.44 | ||||||
| Dividend declared per share |
$ | 0.68 | $ | - | $ | - | ||||||
| Basic shares outstanding (000s) |
142,954 | 145,901 | 152,471 | |||||||||
| Diluted shares outstanding (000s) |
146,410 | 149,316 | 157,195 | |||||||||
See accompanying notes to consolidated financial statements.
- F3 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Consolidated Statements of Comprehensive Income
Years Ended June 30, 2013, 2012 and 2011
(In US$ thousands)
|
Years Ended June 30, |
||||||||||||
|
2013 |
2012 |
2011 |
||||||||||
| Net income |
$ | 307,133 | $ | 254,850 | $ | 226,986 | ||||||
| Other comprehensive income (loss): |
||||||||||||
| Foreign currency translation gain (loss) adjustments |
(144,368 | ) | (87,976 | ) | 238,057 | |||||||
| Comprehensive income |
$ | 162,765 | $ | 166,874 | $ | 465,043 | ||||||
See accompanying notes to consolidated financial statements.
- F4 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Consolidated Statements of Stockholders Equity
Years ended June 30, 2013, 2012 and 2011
(In thousands)
| Common Stock | Additional Paid-in Capital |
Treasury Stock | Retained Earnings |
Accumulated Other Comprehensive Income (Loss) |
Total | |||||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | |||||||||||||||||||||||||||||
| Balance, June 30, 2010 |
160,567 | $ | 605 | $ | 660,185 | (9,222 | ) | $ | (344,505 | ) | $ | 884,876 | $ | 86,375 | $ | 1,287,536 | ||||||||||||||||
| Common stock issued on exercise of options (note 12) |
4,723 | 19 | 87,029 | 87,048 | ||||||||||||||||||||||||||||
| Common stock issued on vesting of restricted stock units, net of shares withheld for tax (note 12) |
189 | 1 | (2,269 | ) | (2,268 | ) | ||||||||||||||||||||||||||
| Common stock issued on employee stock purchase plan (note 12) |
305 | 1 | 8,236 | 8,237 | ||||||||||||||||||||||||||||
| Treasury stock purchases |
(19 | ) | (4,893 | ) | (160,120 | ) | (160,139 | ) | ||||||||||||||||||||||||
| Tax benefit from exercise of options |
14,547 | 14,547 | ||||||||||||||||||||||||||||||
| Stock-based compensation costs |
30,733 | 30,733 | ||||||||||||||||||||||||||||||
| Other comprehensive income |
238,057 | 238,057 | ||||||||||||||||||||||||||||||
| Net income |
226,986 | 226,986 | ||||||||||||||||||||||||||||||
| Dividends declared |
- | - | ||||||||||||||||||||||||||||||
| Balance, June 30, 2011 |
165,784 | $ | 607 | $ | 798,461 | (14,115 | ) | $ | (504,625 | ) | $ | 1,111,862 | $ | 324,432 | $ | 1,730,737 | ||||||||||||||||
| Common stock issued on exercise of options (note 12) |
3,271 | 13 | 56,337 | 56,350 | ||||||||||||||||||||||||||||
| Common stock issued on vesting of restricted stock units, net of shares withheld for tax (note 12) |
329 | 1 | (3,279 | ) | (3,278 | ) | ||||||||||||||||||||||||||
| Common stock issued on employee stock purchase plan (note 12) |
369 | 1 | 8,783 | 8,784 | ||||||||||||||||||||||||||||
| Treasury stock purchases |
(54 | ) | (13,617 | ) | (391,201 | ) | (391,255 | ) | ||||||||||||||||||||||||
| Tax benefit from exercise of options |
8,620 | 8,620 | ||||||||||||||||||||||||||||||
| Stock-based compensation costs |
30,795 | 30,795 | ||||||||||||||||||||||||||||||
| Other comprehensive income |
(87,976 | ) | (87,976 | ) | ||||||||||||||||||||||||||||
| Net income |
254,850 | 254,850 | ||||||||||||||||||||||||||||||
| Dividends declared |
- | - | ||||||||||||||||||||||||||||||
| Balance, June 30, 2012 |
169,753 | $ | 568 | $ | 899,717 | (27,732 | ) | $ | (895,826 | ) | $ | 1,366,712 | $ | 236,456 | $ | 1,607,627 | ||||||||||||||||
| Common stock issued on exercise of options (note 12) |
3,433 | 14 | 65,635 | 65,649 | ||||||||||||||||||||||||||||
| Common stock issued on vesting of restricted stock units, net of shares withheld for tax (note 12) |
529 | 2 | (7,180 | ) | (7,178 | ) | ||||||||||||||||||||||||||
| Common stock issued on employee stock purchase plan (note 12) |
324 | 1 | 10,451 | 10,452 | ||||||||||||||||||||||||||||
| Treasury stock purchases |
(17 | ) | (4,294 | ) | (188,019 | ) | (188,036 | ) | ||||||||||||||||||||||||
| Tax benefit from exercise of options |
18,215 | 18,215 | ||||||||||||||||||||||||||||||
| Stock-based compensation costs |
38,226 | 38,226 | ||||||||||||||||||||||||||||||
| Other comprehensive income |
(144,368 | ) | (144,368 | ) | ||||||||||||||||||||||||||||
| Net income |
307,133 | 307,133 | ||||||||||||||||||||||||||||||
| Dividends declared |
(97,204 | ) | (97,204 | ) | ||||||||||||||||||||||||||||
| Balance, June 30, 2013 |
174,039 | $ | 568 | $ | 1,025,064 | (32,026 | ) | $ | (1,083,845 | ) | $ | 1,576,641 | $ | 92,088 | $ | 1,610,516 | ||||||||||||||||
See accompanying notes to consolidated financial statements.
- F5 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Consolidated Statements of Cash Flows
Years ended June 30, 2013, 2012 and 2011
(In thousands)
| June 30, 2013 |
June 30, 2012 |
June 30, 2011 |
||||||||||
| Cash flows from operating activities: |
||||||||||||
| Net income |
$ | 307,133 | $ | 254,850 | $ | 226,986 | ||||||
| Adjustment to reconcile net income to net cash provided by operating activities: |
||||||||||||
| Depreciation and amortization |
78,280 | 85,856 | 70,616 | |||||||||
| Stock-based compensation costs |
38,157 | 30,586 | 30,809 | |||||||||
| Impairment of long lived assets |
- | - | 2,257 | |||||||||
| Impairment of cost-method investments |
475 | 4,016 | - | |||||||||
| Foreign currency revaluation |
6,258 | (13,652 | ) | (17,261 | ) | |||||||
| Gain on previously held equity interest resulting from business combination |
- | (2,070 | ) | - | ||||||||
| Excess tax benefit from stock-based compensation arrangements |
(18,307 | ) | (8,748 | ) | (14,510 | ) | ||||||
| Changes in operating assets and liabilities, net of effect of acquisitions: |
||||||||||||
| Accounts receivable, net |
(37,267 | ) | (20,293 | ) | (30,799 | ) | ||||||
| Inventories, net |
27,143 | 18,806 | 11,394 | |||||||||
| Prepaid expenses, net deferred income taxes and other current assets |
(28,678 | ) | (37,938 | ) | 12,034 | |||||||
| Accounts payable, accrued expenses and other liabilities |
29,629 | 71,746 | (8,336 | ) | ||||||||
| Net cash provided by operating activities |
402,823 | 383,159 | 283,190 | |||||||||
| Cash flows from investing activities: |
||||||||||||
| Purchases of property, plant and equipment |
(63,579 | ) | (47,135 | ) | (66,609 | ) | ||||||
| Patent registration costs |
(8,203 | ) | (6,972 | ) | (6,431 | ) | ||||||
| Purchases of other intangible assets |
- | (7,000 | ) | - | ||||||||
| Business acquisitions, net of cash acquired |
(5,418 | ) | (53,322 | ) | (22,450 | ) | ||||||
| Investments in cost-method investments |
(2,225 | ) | (4,796 | ) | (2,426 | ) | ||||||
| Proceeds from disposal of cost-method investments |
- | 499 | - | |||||||||
| Proceeds from sale of maturing investment securities |
- | - | 3,950 | |||||||||
| Purchases of foreign currency contracts |
(1,117 | ) | (1,464 | ) | (1,956 | ) | ||||||
| Proceeds from exercise of foreign currency contracts |
2,542 | 18,575 | 19,411 | |||||||||
| Net cash used in investing activities |
(78,000 | ) | (101,615 | ) | (76,511 | ) | ||||||
| Cash flows from financing activities: |
||||||||||||
| Proceeds from issuance of common stock, net |
69,239 | 62,491 | 94,650 | |||||||||
| Excess tax benefit from stock-based compensation arrangements |
18,307 | 8,748 | 14,510 | |||||||||
| Purchases of treasury stock |
(186,258 | ) | (392,743 | ) | (163,342 | ) | ||||||
| Payment of business combination contingent consideration |
(7,790 | ) | - | - | ||||||||
| Proceeds from borrowings |
150,000 | 270,384 | 98,430 | |||||||||
| Repayment of borrowings |
(100,221 | ) | (125,985 | ) | (123,591 | ) | ||||||
| Dividend paid |
(97,204 | ) | - | - | ||||||||
| Net cash used in financing activities |
(153,927 | ) | (177,105 | ) | (79,343 | ) | ||||||
| Effect of exchange rate changes on cash |
(104,389 | ) | (30,165 | ) | 119,155 | |||||||
| Net increase in cash and cash equivalents |
66,507 | 74,274 | 246,491 | |||||||||
| Cash and cash equivalents at beginning of period |
809,541 | 735,267 | 488,776 | |||||||||
| Cash and cash equivalents at end of period |
$ | 876,048 | $ | 809,541 | $ | 735,267 | ||||||
| Supplemental disclosure of cash flow information: |
||||||||||||
| Income taxes paid, net of refunds |
$ | 85,724 | $ | 55,206 | $ | 85,104 | ||||||
| Interest paid |
$ | 6,387 | $ | 4,786 | $ | 1,758 | ||||||
| Fair value of assets acquired, excluding cash |
$ | 5,970 | $ | 24,648 | $ | 18,442 | ||||||
| Liabilities assumed |
(2,278 | ) | (5,056 | ) | (450 | ) | ||||||
| Goodwill on acquisition |
13,876 | 51,798 | 5,758 | |||||||||
| Fair value of contingent consideration |
(12,150 | ) | (6,850 | ) | (800 | ) | ||||||
| Total purchase price, excluding contingent consideration |
5,418 | 64,540 | 22,950 | |||||||||
| Less: Consideration not paid in the current period |
- | (11,218 | ) | (500 | ) | |||||||
| Cash paid for acquisition |
$ | 5,418 | $ | 53,322 | $ | 22,450 | ||||||
See accompanying notes to consolidated financial statements.
- F6 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (1) | Organization and Basis of Presentation |
ResMed Inc. (referred to herein as we, us, our or the Company) is a Delaware corporation formed in March 1994 as a holding company for the ResMed Group. Through our subsidiaries, we design, manufacture and market equipment for the diagnosis and treatment of sleep-disordered breathing and other respiratory disorders, including obstructive sleep apnea. Our manufacturing operations are located in Australia, Singapore, France, Germany, Malaysia and the United States. Major distribution and sales sites are located in the United States, Germany, France, the United Kingdom, Switzerland, Australia, Japan, Norway and Sweden.
| (2) | Summary of Significant Accounting Policies |
| (a) | Basis of Consolidation |
The consolidated financial statements include the accounts of the Company and its wholly owned subsidiaries. All significant inter-company transactions and balances have been eliminated in consolidation.
The preparation of financial statements in conformity with accounting principles generally accepted in the United States requires management estimates and assumptions that affect amounts reported in the consolidated financial statements and accompanying notes. Actual results could differ from managements estimates.
| (b) | Revenue Recognition |
We generally record revenue on product sales at the time of shipment, which is when title transfers to the customer. We do not record revenue on product sales which require customer acceptance until we receive acceptance. We initially defer service revenue received in advance from service contracts and recognize that deferred revenue ratably over the life of the service contract. We initially defer revenue we receive in advance from rental unit contracts and recognize that deferred revenue ratably over the life of the rental contract. Otherwise, we recognize revenue from rental unit contracts ratably over the life of the rental contract. We include in revenue freight charges we bill to customers. We charge all freight-related expenses to cost of sales. Taxes assessed by government authorities that are imposed on and concurrent with revenue-producing transactions, such as sales and value added taxes, are excluded from revenue.
We do not recognize revenues to the extent that we offer a right of return or other recourse with respect to the sale of our products, other than returns for product defects or other warranty claims, nor do we recognize revenues if we offer variable sale prices for subsequent events or activities. However, as part of our sales processes we may provide upfront discounts for large orders, one-time special pricing to support new product introductions, sales rebates for centralized purchasing entities or price-breaks for regular order volumes. We record the costs of all such programs as an adjustment to revenue. Our products are predominantly therapy-based equipment and require no installation. Therefore, we have no installation obligations.
| (c) | Cash and Cash Equivalents |
Cash equivalents include certificates of deposit and other highly liquid investments and we state them at cost, which approximates market. We consider investments with original maturities of 90 days or less to be cash equivalents for purposes of the consolidated statements of cash flows.
Our cash and cash equivalents balance at June 30, 2013, include $90.5 million in cash which is subject to a notice period of 90 days. These cash balances earn interest rates above normal term deposit rates otherwise available and are held at highly rated financial institutions.
- F7 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (2) | Summary of Significant Accounting Policies, Continued |
| (d) | Inventories |
We state inventories at the lower of cost (determined principally by the first-in, first-out method) or net realizable value. We include material, labor and manufacturing overhead costs in finished goods and work-in-process inventories. We review and provide for any product obsolescence in our manufacturing and distribution operations by assessing throughout the year individual products and components (based on estimated future usage and sales).
| (e) | Property, Plant and Equipment |
We record property, plant and equipment, including rental equipment at cost. We compute depreciation expense using the straight-line method over the estimated useful lives of the assets. Useful lives are generally two to ten years except for buildings which are depreciated over an estimated useful life of 40 years and leasehold improvements, which we amortize over the lease term. We charge maintenance and repairs to expense as we incur them.
| (f) | Intangible Assets |
We capitalize the registration costs for new patents and amortize the costs over the estimated useful life of the patent, which is generally five years. If a patent is superseded or a product is retired, any unamortized costs are written off immediately.
We amortize all of our other intangible assets on a straight-line basis over their estimated useful lives, which range from two to nine years. We evaluate the recoverability of intangible assets periodically and take into account events or circumstances that warrant revised estimates of useful lives or that indicate that impairment exists. We have not identified any impairment of intangible assets during any of the periods presented.
| (g) | Goodwill |
We conducted our annual review for goodwill impairment during the final quarter of fiscal 2013 using a quantitative assessment. In conducting our review of goodwill impairment, we identified 8 reporting units, being components of our operating segment. The fair value for each reporting unit was determined based on estimated discounted cash flows. Our goodwill impairment review involved a two-step process as follows:
| Step 1 |
Compare the fair value for each reporting unit to its carrying value, including goodwill. For each reporting unit where the carrying value, including goodwill, exceeds the reporting units fair value, move on to step 2. If a reporting units fair value exceeds the carrying value, no further work is performed and no impairment charge is necessary. | |
| Step 2 |
Allocate the fair value of the reporting unit to its identifiable tangible and non-goodwill intangible assets and liabilities. This will derive an implied fair value for the goodwill. Then, compare the implied fair value of the reporting units goodwill with the carrying amount of the reporting units goodwill. If the carrying amount of the reporting units goodwill is greater than the implied fair value of its goodwill, an impairment loss must be recognized for the excess. | |
The results of Step 1 of our annual review indicated that no impaired goodwill exists as the fair value for each reporting unit significantly exceeded its carrying value.
- F8 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (2) | Summary of Significant Accounting Policies, Continued |
| (h) | Foreign Currency |
The consolidated financial statements of our non-U.S. subsidiaries, whose functional currencies are other than the U.S. dollar, are translated into U.S. dollars for financial reporting purposes. We translate assets and liabilities of non-U.S. subsidiaries whose functional currencies are other than the U.S. dollar at period end exchange rates, but translate revenue and expense transactions at average exchange rates for the period. We recognize cumulative translation adjustments as part of comprehensive income, as detailed in the Consolidated Statements of Comprehensive Income, and include those adjustments in accumulated other comprehensive income in the consolidated balance sheets until such time the relevant subsidiary is sold or substantially or completely liquidated. We reflect gains and losses on transactions denominated in other than the functional currency of an entity in our results of operations.
| (i) | Research and Development |
We record all research and development expenses in the period we incur them.
| (j) | Financial Instruments |
The carrying value of financial instruments, such as cash and cash equivalents, accounts receivable and accounts payable, approximate their fair value because of their short-term nature. The carrying value of long-term debt approximates its fair value as the principal amounts outstanding are subject to variable interest rates that are based on market rates which are regularly reset. Foreign currency option contracts are marked to market and therefore reflect their fair value. We do not hold or issue financial instruments for trading purposes.
The fair value of financial instruments is the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date.
| (k) | Foreign Exchange Risk Management |
We enter into various types of foreign exchange contracts in managing our foreign exchange risk, including derivative financial instruments encompassing forward exchange contracts and foreign currency options.
The purpose of our foreign currency hedging activities is to protect us from adverse exchange rate fluctuations with respect to net cash movements resulting from the sales of products to foreign customers and Australian and Singapore manufacturing activities. We enter into foreign currency option contracts to hedge anticipated sales and manufacturing costs, principally denominated in Australian and Singapore dollars, and Euros. The terms of such foreign currency option contracts generally do not exceed three years.
We have determined our hedge program to be a non-effective hedge as defined. We record the foreign currency derivatives portfolio at fair value and include it in other assets and accrued expenses in our consolidated balance sheets. We do not offset the fair value amounts recognized for foreign currency derivatives. We classify purchases of foreign currency derivatives and proceeds received from the exercise of foreign currency derivatives as an investing activity within our consolidated statements of cash flows.
We record all movements in the fair value of the foreign currency derivatives within other income, net in our consolidated statements of income.
- F9 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (2) | Summary of Significant Accounting Policies, Continued |
| (l) | Income Taxes |
We account for income taxes under the asset and liability method. We recognize deferred tax assets and liabilities for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases. We measure deferred tax assets and liabilities using the enacted tax rates we expect to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that includes the enactment date.
| (m) | Allowance for Doubtful Accounts |
We maintain an allowance for doubtful accounts for estimated losses resulting from the inability of our customers to make required payments, which results in bad debt expense. We determine the adequacy of this allowance by periodically evaluating individual customer receivables, considering a customers financial condition, credit history and current economic conditions. We are also contingently liable, within certain limits, in the event of a customer default, to several independent leasing companies in connection with customer leasing programs. We monitor the collection status of these installment receivables and provide for estimated losses separately under accrued expenses within our consolidated balance sheets based upon our historical collection experience with such receivables and a current assessment of our credit exposure.
| (n) | Impairment of Long-Lived Assets |
We periodically evaluate the carrying value of long-lived assets to be held and used, including certain identifiable intangible assets, when events and circumstances indicate that the carrying amount of an asset may not be recovered. Recoverability of assets to be held and used is measured by a comparison of the carrying amount of an asset to future net cash flows expected to be generated by the asset. If assets are considered to be impaired, we recognize as the impairment the amount by which the carrying amount of the assets exceeds the fair value of the assets. We report assets to be disposed of at the lower of the carrying amount or fair value less costs to sell.
During the year ended June 30, 2013, 2012 and 2011, we recognized an impairment charge of $Nil, $Nil and $2.3 million, respectively, relating to impaired long-lived assets that were no longer in use. The impairment charge related to the long-lived assets, in fiscal year 2011, was recorded in cost of sales in our consolidated statements of income.
| (3) | New Accounting Pronouncements |
In June 2011, the FASB issued authoritative guidance with respect to the presentation of other comprehensive income in financial statements. The main provisions of the standard provide that an entity that reports other comprehensive income has the option to present comprehensive income in either a single statement or in a two-statement approach. A single statement must present the components of net income and total net income, the components of other comprehensive income and total other comprehensive income, and a total for comprehensive income. In the two-statement approach, an entity must present the components of net income and total net income in the first statement, followed by a financial statement that presents the components of other comprehensive income, a total for other comprehensive income, and a total for comprehensive income. The adoption of this standard in fiscal year 2013 affected the presentation of our other comprehensive income but not our financial position or results of operations.
- F10 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (4) | Earnings Per Share |
We compute basic earnings per share by dividing the net income available to common stockholders by the weighted average number of shares of common stock outstanding. For purposes of calculating diluted earnings per share, the denominator includes both the weighted average number of shares of common stock outstanding and the number of dilutive common stock equivalents such as stock options and restricted stock units.
The weighted average number of outstanding stock options and restricted stock units not included in the computation of diluted earnings per share were 236,000, 1,336,000 and 651,000 for the years ended June 30, 2013, 2012 and 2011, respectively, as the effect would have been anti-dilutive.
Basic and diluted earnings per share for the years ended June 30, 2013, 2012 and 2011 are calculated as follows (in thousands except per share data):
| 2013 | 2012 | 2011 | ||||||||||
| Numerator: |
||||||||||||
| Net Income, used in calculating diluted earnings per share |
$ | 307,133 | $ | 254,850 | $ | 226,986 | ||||||
| Denominator: |
||||||||||||
| Basic weighted-average common shares outstanding |
142,954 | 145,901 | 152,471 | |||||||||
| Effect of dilutive securities: |
||||||||||||
| Stock options and restricted stock units |
3,456 | 3,415 | 4,724 | |||||||||
| Diluted weighted average shares |
146,410 | 149,316 | 157,195 | |||||||||
| Basic earnings per share |
$ | 2.15 | $ | 1.75 | $ | 1.49 | ||||||
| Diluted earnings per share |
$ | 2.10 | $ | 1.71 | $ | 1.44 | ||||||
| (5) | Inventories |
Inventories were comprised of the following as of June 30, 2013 and June 30, 2012 (in thousands):
| 2013 | 2012 | |||||||
| Raw materials |
$ | 46,841 | $ | 65,518 | ||||
| Work in progress |
1,990 | 1,692 | ||||||
| Finished goods |
97,016 | 107,141 | ||||||
| Total inventories |
$ | 145,847 | $ | 174,351 | ||||
- F11 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (6) | Property, Plant and Equipment, net |
Property, plant and equipment, net is comprised of the following as of June 30, 2013 and June 30, 2012 (in thousands):
| 2013 | 2012 | |||||||
| Machinery and equipment |
$ | 165,782 | $ | 158,542 | ||||
| Computer equipment |
109,657 | 102,143 | ||||||
| Furniture and fixtures |
40,706 | 41,818 | ||||||
| Vehicles |
3,282 | 3,046 | ||||||
| Clinical, demonstration and rental equipment |
102,304 | 94,176 | ||||||
| Leasehold improvements |
28,466 | 25,220 | ||||||
| Land |
61,091 | 65,928 | ||||||
| Buildings |
260,857 | 277,743 | ||||||
| 772,145 | 768,616 | |||||||
| Accumulated depreciation and amortization |
(360,712 | ) | (334,253 | ) | ||||
| Property, plant and equipment, net |
$ | 411,433 | $ | 434,363 | ||||
| (7) | Goodwill and Other Intangible Assets, net |
Goodwill
Changes in the carrying amount of goodwill for the years ended June 30, 2013 and June 30, 2012 (in thousands):
| 2013 | 2012 | |||||||
| Balance at the beginning of the period |
$ | 256,209 | $ | 235,487 | ||||
| Business acquisition (note 22) |
13,876 | 51,798 | ||||||
| Foreign currency translation adjustments |
4,744 | (31,076 | ) | |||||
| Balance at the end of the period |
$ | 274,829 | $ | 256,209 | ||||
As at June 30, 2013 we have not recorded any accumulated goodwill impairments.
- F12 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (7) | Goodwill and Other Intangible Assets, net, Continued |
Other Intangible Assets
Other intangibles, net are comprised of the following as of June 30, 2013 and June 30, 2012:
| 2013 | 2012 | |||||||
| Developed/core product technology |
$ | 72,698 | $ | 67,263 | ||||
| Accumulated amortization |
(45,492 | ) | (39,036 | ) | ||||
| Developed/core product technology, net |
27,206 | 28,227 | ||||||
| Trade names |
2,662 | 2,628 | ||||||
| Accumulated amortization |
(2,491 | ) | (2,276 | ) | ||||
| Trade names, net |
171 | 352 | ||||||
| Non compete agreements |
2,068 | 2,321 | ||||||
| Accumulated amortization |
(1,265 | ) | (886 | ) | ||||
| Non compete agreements, net |
803 | 1,435 | ||||||
| Customer relationships |
22,291 | 22,783 | ||||||
| Accumulated amortization |
(17,095 | ) | (14,097 | ) | ||||
| Customer relationships, net |
5,196 | 8,686 | ||||||
| Patents |
59,962 | 58,389 | ||||||
| Accumulated amortization |
(43,699 | ) | (42,262 | ) | ||||
| Patents, net |
16,263 | 16,127 | ||||||
| Total other intangibles, net |
$ | 49,639 | $ | 54,827 | ||||
Intangible assets consist of developed/core product technology, trade names, non-compete agreements, customer relationships and patents, and we amortize them over the estimated useful life of the assets, generally between two and nine years. There are no expected residual values related to these intangible assets.
Refer to Note 22 of the consolidated financial statements for further details of acquisitions made during the year.
Amortization expense related to identifiable intangible assets, including patents, for the year ended June 30, 2013 was $17.0 million. Estimated annual amortization expense for the years ending June 30, 2014 through June 30, 2018, is shown below (in thousands):
| Fiscal Year | Amortization expense | |||
| 2014 |
$ | 14,944 | ||
| 2015 |
12,233 | |||
| 2016 |
10,542 | |||
| 2017 |
6,766 | |||
| 2018 |
3,183 | |||
- F13 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (8) | Cost-Method Investments |
The aggregate carrying amount of our cost-method investments at June 30, 2013 and June 30, 2012, was $4.0 million and $2.3 million, respectively. During the year ended June 30, 2012 we remeasured a previously held equity interest to its acquisition date fair value as a result of acquiring the remaining interest as part of a business combination and recognized a gain of $2.1 million in other income, net within our consolidated statements of income.
We periodically evaluate the carrying value of our cost-method investments, when events and circumstances indicate that the carrying amount of an asset may not be recovered. We determine the fair value of our cost-method investments to evaluate whether impairment losses shall be recorded using Level 3 inputs. These investments include our holdings in privately held service and research companies that are not exchange traded and therefore not supported with observable market prices. However, these investments are valued by reference to their net asset values which can be market supported and unobservable inputs including future cash flows. During the year ended June 30, 2013 and 2012, we recognized $0.5 million and $4.0 million, respectively, of impairment losses related to our cost-method investments. The expense associated with this impairment has been included in other income, net within our consolidated statements of income. We based these impairment losses on our determination that the declines in the fair value of these investments were other-than temporary. We have determined, after the impairment charge, that the fair value of our remaining investments exceed their carrying values.
The following table shows a reconciliation of the changes in our cost-method investments during the years ended June 30, 2013 and June 30, 2012 (in thousands):
| 2013 | 2012 | |||||||
| Balance at the beginning of the period |
$ | 2,250 | $ | 4,264 | ||||
| Investments |
2,225 | 4,796 | ||||||
| Elimination due to acquisition of entity |
- | (2,261 | ) | |||||
| Disposals |
- | (455 | ) | |||||
| Impairment of cost-method investments |
(475 | ) | (4,016 | ) | ||||
| Foreign currency translation adjustments |
- | (78 | ) | |||||
| Balance at the end of the period |
$ | 4,000 | $ | 2,250 | ||||
| (9) | Accrued Expenses |
Accrued expenses at June 30, 2013 and June 30, 2012 consist of the following (in thousands):
| 2013 | 2012 | |||||||
| Product warranties |
$ | 16,011 | $ | 17,018 | ||||
| Consulting and professional fees |
7,360 | 7,327 | ||||||
| Value added taxes and other taxes due |
13,061 | 14,756 | ||||||
| Employee related costs |
70,387 | 67,668 | ||||||
| Marketing and promotional programs |
1,262 | 2,125 | ||||||
| Business acquisition contingent consideration |
7,779 | 5,024 | ||||||
| Hedging instruments |
9,007 | - | ||||||
| Other |
12,807 | 13,463 | ||||||
| $ | 137,674 | $ | 127,381 | |||||
- F14 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (10) | Product Warranties |
Estimated future warranty costs related to certain products are charged to operations in the period in which the related revenue is recognized. We include the liability for warranty costs in accrued expenses in our consolidated balance sheets.
Changes in the liability for product warranty for the years ended June 30, 2013 and 2012 are as follows (in thousands):
| 2013 | 2012 | |||||||
| Balance at the beginning of the period |
$ | 17,018 | $ | 19,032 | ||||
| Warranty accruals for the period |
11,426 | 11,027 | ||||||
| Warranty costs incurred for the period |
(10,538 | ) | (12,077 | ) | ||||
| Foreign currency translation adjustments |
(1,895 | ) | (964 | ) | ||||
| Balance at the end of the period |
$ | 16,011 | $ | 17,018 | ||||
| (11) | Long-term Debt |
Long-term debt at June 30, 2013 and June 30, 2012 consists of the following (in thousands):
| June 30, 2013 |
June 30, 2012 |
|||||||
| Current long-term debt |
$ | 300,017 | $ | 52 | ||||
| Non-current long-term debt |
769 | 250,783 | ||||||
| Total long-term debt |
$ | 300,786 | $ | 250,835 | ||||
Credit Facility
During the year ended June 30, 2011, we entered into a credit agreement with lenders, including Union Bank, N.A., as Administrative Agent, Swing Line Lender and L/C Issuer, HSBC Bank USA, National Association, as Syndication Agent and Union Bank, N.A., HSBC Bank USA, National Association, Commonwealth Bank of Australia and Wells Fargo Bank, N.A. The credit agreement provides a $300 million three-year revolving credit facility, with an uncommitted option to increase the credit facility by an additional $100 million. The credit facility also includes a $10 million sublimit for letters of credit. The credit facility terminates on February 10, 2014, at which time all unpaid principal and interest under the loans must be repaid. The outstanding principal amount due under the credit facility will bear interest at a rate equal to, at our option, either (i) LIBOR plus 1.5% to 2.0% (depending on the applicable leverage ratio) or (ii) a base rate, as defined in the credit agreement, plus 0.5% to 1.0% (depending on the applicable leverage ratio). At June 30, 2013, the interest rate that was being charged on the outstanding principal amount was 1.7%. Commitment fees of 0.25% to 0.375% (depending on the applicable leverage ratio) apply on the unused portion of the credit facility. When we executed the credit agreement, we used a portion of the credit facilitys initial funding proceeds to repay the outstanding balance under our previously existing revolving credit facility with Union Bank, N.A., which was then terminated.
Our obligations under the credit agreement are secured by (a) the corporate stock we hold in our subsidiaries ResMed Corp. and ResMed Motor Technologies Inc. (ResMed Motor), and (b) up to 65% of the ownership interests we hold in our subsidiary ResMed EAP Holdings LLC (ResMed EAP). Our
- F15 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (11) | Long-term Debt, Continued |
obligations under the credit agreement are also guaranteed by our subsidiaries ResMed Corp and ResMed Motor. The credit agreement contains customary covenants, including certain financial covenants and an obligation that we maintain certain financial ratios, including a maximum ratio of Funded Debt to EBITDA (each as defined in the credit agreement), an interest coverage ratio and a maximum amount of annual capital expenditures. The entire principal amount of the credit facility and any accrued but unpaid interest may be declared immediately due and payable if an event of default occurs. Events of default include failure to make payments when due, a default in the performance of any covenants in the credit agreement or related documents or certain changes of control of us or our subsidiaries ResMed Corp, ResMed Motor, ResMed Limited, ResMed Holdings Ltd/LLC or ResMed EAP.
On January 25, 2012, we entered into a first amendment to the credit agreement. The amendment increases, from $300 million to $400 million, the maximum principal amount that can be borrowed on a revolving basis under the credit agreement, subject to customary conditions.
At June 30, 2013, there was $300.0 million outstanding under the credit agreement.
| (12) | Stockholders Equity |
Common Stock. On August 24, 2011, our board of directors approved a new share repurchase program, authorizing us to acquire up to an aggregate of 20.0 million shares of ResMed Inc. common stock. The program allows us to repurchase shares of our common stock from time to time for cash in the open market, or in negotiated or block transactions, as market and business conditions warrant. This program canceled and replaced our previous share repurchase program authorized on May 27, 2009 pursuant to which we had repurchased 10.0 million shares. These were in addition to the 6.6 million shares repurchased under an earlier program authorized on June 6, 2002. The new program authorizes us to purchase in addition to the shares we repurchased under our previous programs. There is no expiration date for this program. All share repurchases since August 24, 2011 have been executed in accordance with this program.
During the fiscal years 2013 and 2012, we repurchased 4.3 million and 13.6 million shares, respectively, at a cost of $188.0 million and $391.2 million, respectively. At June 30, 2013, we have repurchased a total of 32.0 million shares at a cost of $1,083.8 million. Shares that are repurchased are classified as treasury stock pending future use and reduce the number of shares outstanding used in calculating earnings per share. At June 30, 2013, 4.5 million additional shares can be repurchased under the approved share repurchase program.
Preferred Stock. In April 1997, our board of directors authorized 2,000,000 shares of $0.01 par value preferred stock. No such shares were issued or outstanding at June 30, 2013.
Stock Options and Restricted Stock Units. We have granted stock options and restricted stock units to personnel, including officers and directors, in accordance with the ResMed Inc. 2009 Incentive Award Plan (the 2009 Plan). These options and restricted stock units have expiration dates of seven years from the date of grant and vest over one or four years. We have granted the options with an exercise price equal to the market value as determined at the date of grant.
The maximum number of shares of our common stock authorized for issuance under the 2009 Plan is 35.5 million. The number of securities remaining available for future issuance under the 2009 Plan at June 30, 2013 is 10.6 million. The number of shares of our common stock available for issuance under the 2009 Plan will be reduced by (i) three shares for each one share of common stock delivered in settlement of any full-value award, which is any award other than a stock option, stock appreciation right or other award for which the holder pays the intrinsic value and (ii) one share for each share of common stock delivered in settlement of all other awards. The maximum number of shares, which may be subject to awards granted
- F16 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (12) | Stockholders Equity, Continued |
under the 2009 Plan to any individual during any calendar year, may not exceed 3 million shares of our common stock (except in a participants initial year of hiring up to 4.5 million shares of our common stock may be granted).
At June 30, 2013, there was $77.6 million in unrecognized compensation costs related to unvested stock-based compensation arrangements. This is expected to be recognized over a weighted average period of 2.5 years. The aggregate intrinsic value of the stock-based compensation arrangements outstanding and exercisable at June 30, 2013 was $260.6 million and $121.9 million, respectively. The aggregate intrinsic value of the options exercised during the fiscal years 2013, 2012 and 2011 was $77.8 million, $45.3 million and $66.4 million, respectively.
The following table summarizes option activity during the year ended June 30, 2013:
| Weighted Average Exercise Price |
Weighted Average Remaining Term to Vest in Years |
|||||||||||
| Outstanding at beginning of period |
9,363,720 | $ | 20.52 | 3.3 | ||||||||
| Granted |
444,766 | 41.11 | ||||||||||
| Exercised |
(3,432,800 | ) | 19.14 | |||||||||
| Forfeited |
(59,550 | ) | 23.87 | |||||||||
|
Outstanding at end of period |
6,316,136 | $ | 22.68 | 2.3 | ||||||||
| Exercise price range of granted options |
31.61-43.63 | |||||||||||
| Options exercisable at end of period |
4,727,408 | $ | 19.35 | |||||||||
The following table summarizes the activity of restricted stock units during year ended June 30, 2013:
| Weighted Average Grant- Date Fair Value |
Weighted Average Remaining Term to Vest in Years |
|||||||||||
| Outstanding at beginning of period |
2,160,873 | $ | 29.13 | 1.6 | ||||||||
| Granted |
1,259,599 | 37.72 | ||||||||||
| Vested* |
(699,388 | ) | 29.00 | |||||||||
| Forfeited |
(87,677 | ) | 30.15 | |||||||||
|
Outstanding at end of period |
2,633,407 | $ | 33.25 | 1.4 | ||||||||
* Includes 170,606 shares netted for tax.
Employee Stock Purchase Plan (the ESPP). At the annual meeting of our stockholders on November 15, 2012, our stockholders approved an amendment to the ESPP to increase the number of shares of common stock that may be issued or transferred pursuant to awards under the ESPP by 2.0 million shares, from 2.2 million shares to 4.2 million shares. Under the ESPP, we offer participants the right to purchase shares of our common stock at a discount during successive offering periods. Each offering period under the ESPP will be for a period of time determined by the board of directors compensation committee of no less than 3 months and no more than 27 months. The purchase price for our common stock under the ESPP will be the lower of 85% of the fair market value of our common stock on the date of grant or 85% of the fair market value of our common stock on the date of purchase. An individual participant cannot subscribe for more than $25,000 in common stock during any calendar year. At June 30, 2013, the number of shares remaining available for future issuance under the ESPP is 2.1 million shares.
- F17 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (12) | Stockholders Equity, Continued |
During fiscal years 2013 and 2012, we issued 324,000 and 369,000 shares to our employees in two offerings and we recognized $2.8 million and $2.8 million, respectively, of stock compensation expense associated with the ESPP.
The following table summarizes the total stock-based compensation costs incurred and the associated tax benefit recognized during the year ended June 30, 2013, 2012 and 2011 (in thousands):
| 2013 | 2012 | 2011 | ||||||||||
| Cost of sales capitalized as part of inventory |
$ | 2,484 | $ | 1,749 | $ | 1,496 | ||||||
| Selling, general and administrative expenses |
30,552 | 25,201 | 26,163 | |||||||||
| Research and development expenses |
5,121 | 3,636 | 3,150 | |||||||||
| Stock-based compensation costs |
38,157 | 30,586 | 30,809 | |||||||||
| Tax benefit |
(11,960 | ) | (8,421 | ) | (9,474 | ) | ||||||
| Stock-based compensation costs, net of tax benefit |
$ | 26,197 | $ | 22,165 | $ | 21,335 | ||||||
| (13) | Other, net |
Other, net, in the consolidated statements of income is comprised of the following for the years ended June 30, 2013, 2012 and 2011 (in thousands):
| 2013 | 2012 | 2011 | ||||||||||
| Gain (loss) on foreign currency transactions and hedging, net |
$ | (1,902 | ) | $ | 9,766 | $ | 10,619 | |||||
| Impairment of cost method investments |
(475 | ) | (4,016 | ) | - | |||||||
| Gain on re-measurement of equity interest |
- | 2,070 | - | |||||||||
| Other |
186 | 638 | 121 | |||||||||
| $ | (2,191 | ) | $ | 8,458 | $ | 10,740 | ||||||
| (14) | Income Taxes |
Income before income taxes for the years ended June 30, 2013, 2012 and 2011, was taxed under the following jurisdictions (in thousands):
| 2013 | 2012 | 2011 | ||||||||||
| U.S. |
$ | 2,120 | $ | 8,542 | $ | (1,419 | ) | |||||
| Non-U.S. |
382,999 | 323,403 | 305,126 | |||||||||
| $ | 385,119 | $ | 331,945 | $ | 303,707 | |||||||
- F18 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (14) | Income Taxes, Continued |
The provision for income taxes is presented below (in thousands):
| 2013 | 2012 | 2011 | ||||||||||
| Current: Federal |
$ | 42,065 | $ | 16,201 | $ | 10,461 | ||||||
| State |
888 | 2,163 | 1,435 | |||||||||
| Non-U.S. |
53,713 | 71,353 | 61,469 | |||||||||
| 96,666 | 89,717 | 73,365 | ||||||||||
| Deferred: Federal |
(1,345 | ) | (352 | ) | 217 | |||||||
| State |
(264 | ) | (178 | ) | (226 | ) | ||||||
| Non-U.S. |
(17,071 | ) | (12,092 | ) | 3,365 | |||||||
| (18,680 | ) | (12,622 | ) | 3,356 | ||||||||
| Provision for income taxes |
$ | 77,986 | $ | 77,095 | $ | 76,721 | ||||||
The provision for income taxes differs from the amount of income tax determined by applying the applicable U.S. federal income tax rate of 35% to pretax income as a result of the following (in thousands):
| 2013 | 2012 | 2011 | ||||||||||
| Taxes computed at statutory U.S. rate |
$ | 134,792 | $ | 116,181 | $ | 106,297 | ||||||
| Increase (decrease) in income taxes resulting from: |
||||||||||||
| State income taxes, net of U.S. tax benefit |
257 | 1,206 | 1,060 | |||||||||
| Non-deductible expenses |
1,667 | 2,260 | 1,113 | |||||||||
| Research and development credit |
(4,920 | ) | (4,210 | ) | (7,463 | ) | ||||||
| Tax effect of dividends |
72,304 | 33,656 | 40,038 | |||||||||
| Change in valuation allowance |
1,852 | 1,645 | (2,748 | ) | ||||||||
| Effect of non-U.S. tax rates |
(97,420 | ) | (57,252 | ) | (38,269 | ) | ||||||
| Foreign tax credits |
(34,729 | ) | (18,179 | ) | (25,738 | ) | ||||||
| Stock-based compensation expense |
3,167 | 2,558 | 2,027 | |||||||||
| Other |
1,016 | (770 | ) | 404 | ||||||||
| $ | 77,986 | $ | 77,095 | $ | 76,721 | |||||||
- F19 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (14) | Income Taxes, Continued |
We classify deferred tax assets and liabilities as current or non-current according to the related asset or liabilitys classification. The components of our deferred tax assets and liabilities at June 30, 2013 and 2012 are as follows (in thousands):
| 2013 | 2012 | |||||||
| Deferred tax assets: |
||||||||
| Employee liabilities |
$ | 10,602 | $ | 10,748 | ||||
| Inventories |
10,398 | 9,811 | ||||||
| Provision for warranties |
4,691 | 4,334 | ||||||
| Provision for doubtful debts |
2,820 | 1,960 | ||||||
| Net operating loss carryforwards |
15,432 | 8,363 | ||||||
| Capital loss carryover |
2,015 | 1,247 | ||||||
| Stock-based compensation expense |
17,460 | 17,355 | ||||||
| Unrealized foreign exchange losses |
1,912 | - | ||||||
| Other |
4,070 | 1,337 | ||||||
| 69,400 | 55,155 | |||||||
| Less valuation allowance |
(9,671 | ) | (5,910 | ) | ||||
| Deferred tax assets |
59,729 | 49,245 | ||||||
| Deferred tax liabilities: |
||||||||
| Unrealized foreign exchange gains |
- | (5,369 | ) | |||||
| Property, plant and equipment |
(1,560 | ) | (1,573 | ) | ||||
| Goodwill and other intangibles |
(10,086 | ) | (9,129 | ) | ||||
| Deferred tax liabilities |
(11,646 | ) | (16,071 | ) | ||||
| Net deferred tax asset |
$ | 48,083 | $ | 33,174 | ||||
We reported the net deferred tax assets and liabilities in our consolidated balance sheets at June 30, 2013 and 2012 as follows (in thousands):
| 2013 | 2012 | |||||||
| Current deferred tax asset |
$ | 38,552 | $ | 19,590 | ||||
| Non-current deferred tax asset |
20,053 | 23,500 | ||||||
| Current deferred tax liability |
(627 | ) | (1,073 | ) | ||||
| Non-current deferred tax liability |
(9,895 | ) | (8,843 | ) | ||||
| Net deferred tax asset |
$ | 48,083 | $ | 33,174 | ||||
At June 30, 2013, we had $12.1 million of U.S. state net operating loss carryforwards and $62.6 million of non-U.S. net operating loss carryforwards, which expire in various years through 2033 or carry forward indefinitely.
The valuation allowance at June 30, 2013 relates to a provision for uncertainty as to the utilization of net operating loss carryforwards for certain non-U.S. countries of $7.1 million and U.S. and non-U.S. capital loss and other items of $2.6 million. We believe that it is more likely than not that the benefits of deferred tax assets, net of any valuation allowance, will be realized.
- F20 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (14) | Income Taxes, Continued |
A substantial portion of the Companys manufacturing operations and administrative functions in Malaysia and Singapore operate under various tax holidays and tax incentive programs that will expire in whole or in part at various dates through June 30, 2020. Certain of the holidays may be extended if specific conditions are met. The net impact of these tax holidays and tax incentives program was to increase the Companys net earnings by $24.3 million ($0.17 per diluted share) for the year ended June 30, 2013 and $3.9 million ($0.03 per diluted share) for the year ended June 30, 2012.
At June 30, 2013, applicable U.S. federal income taxes and foreign withholding taxes have not been provided on the accumulated earnings of foreign subsidiaries that are expected to be permanently reinvested. The total amount of these undistributed earnings at June 30, 2013 amounted to approximately $1.6 billion. If these earnings had not been permanently reinvested, deferred taxes of approximately $409 million would have been recognized in the consolidated financial statements.
In accounting for uncertainty in income taxes, we recognize a tax benefit in the financial statements for an uncertain tax position only if managements assessment is that the position is more likely than not (i.e., a likelihood greater than 50 percent) to be allowed by the tax jurisdiction based solely on the technical merits of the position. The term tax position refers to a position in a previously filed tax return or a position expected to be taken in a future tax return that is reflected in measuring current or deferred income tax assets and liabilities for annual periods.
The Company recognizes interest and penalties related to unrecognized tax benefits within the income tax expense line in the accompanying consolidated statements of income. Accrued interest and penalties are included within the related tax liability line in the consolidated balance sheets.
The following table indicates the changes to our unrecognized tax benefits for the year ended June 30, 2013 and June 30, 2012 (in thousands). The term unrecognized tax benefits, or UTB, refers to the differences between a tax position taken or expected to be taken in a tax return and the benefit measured and recognized in the consolidated financial statements.
| 2013 | 2012 | |||||||
| Gross UTB balance at beginning of period |
$ | 3,868 | $ | 4,284 | ||||
| Additions for tax positions of prior years |
163 | 138 | ||||||
| Reductions due to lapse of applicable statute of limitations |
- | - | ||||||
| Foreign exchange movement |
74 | (554 | ) | |||||
| Gross UTB balance at end of period |
$ | 4,105 | $ | 3,868 | ||||
Included in the balance at June 30, 2013, are tax positions of $3.5 million that, if recognized, would affect our effective tax rate. As of June 30, 2013, we have accrued approximately $1.8 million ($1.3 million, net of tax benefit) for interest and penalties related to uncertain tax positions in the income taxes payable balance on the consolidated balance sheet.
We file numerous consolidated and separate income tax returns in the U.S. federal jurisdiction and in many state and foreign jurisdictions. We are no longer subject to U.S. federal income tax examination for tax years prior to fiscal year 2010, and no longer subject to state income tax examinations for the tax years prior to fiscal year 2009. With few exceptions, we are no longer subject to foreign income tax examinations for fiscal years before 2006.
Within the next 12 months, we do not anticipate a decrease in the unrecognized tax benefit or any other significant changes within our tax reserves.
- F21 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (15) | Segment Information |
We operate solely in the sleep-disordered breathing sector of the respiratory medicine industry. We therefore believe that, given the single market focus of our operations and the inter-dependence of our products, we operate as a single operating segment. We assess performance and allocate resources on the basis of a single operating entity.
Sales of flow generators for the years ended June 30, 2013, June 30, 2012 and June 30, 2011 were $823.5 million, $736.6 million and $699.3 million, respectively. Sales of mask systems, motors and other accessories for the years ended June 30, 2013, June 30, 2012 and June 30, 2011 were $690.9 million, $631.9 million and $543.9 million, respectively. Financial information by geographic area for the years ended June 30, 2013, 2012 and 2011, is summarized below (in thousands):
| Revenue from external sources | Long lived assets | |||||||||||||||||||||||
| for the years ended June 30, | at June 30, | |||||||||||||||||||||||
| 2013 | 2012 | 2011 | 2013 | 2012 | 2011 | |||||||||||||||||||
| North and Latin America |
$ | 851,608 | $ | 749,039 | $ | 662,240 | $ | 136,984 | $ | 142,853 | $ | 148,840 | ||||||||||||
| Germany |
195,091 | 181,421 | 171,394 | 24,855 | 27,489 | 26,320 | ||||||||||||||||||
| France |
138,891 | 139,286 | 137,330 | 6,282 | 4,870 | 6,276 | ||||||||||||||||||
| Australia |
35,503 | 35,654 | 34,975 | 222,814 | 245,273 | 268,695 | ||||||||||||||||||
| Rest of the World |
293,364 | 263,115 | 237,209 | 20,498 | 13,878 | 11,976 | ||||||||||||||||||
| Total |
$ | 1,514,457 | $ | 1,368,515 | $ | 1,243,148 | $ | 411,433 | $ | 434,363 | $ | 462,107 | ||||||||||||
Long-lived assets of geographic areas are those assets used in our operations in each geographical area, and excludes goodwill, other intangible assets, and deferred tax assets.
| (16) | Stock-based Employee Compensation |
We measure the compensation expense of all stock-based awards at fair value on the grant date. We estimate the fair value of stock options and purchase rights granted under the ESPP using the Black-Scholes valuation model. The fair value of restricted stock units is equal to the market value of the underlying shares as determined at the grant date less the fair value of dividends that holders are not entitled to, during the vesting period. We recognize the fair value as compensation expense using the straight-line method over the service period for awards expected to vest.
We estimate the fair value of stock options granted under our stock option plans and purchase rights granted under the ESPP using the following assumptions:
| Fiscal Year Ended June 30, | ||||||||
| 2013 | 2012 | |||||||
| Stock options: |
||||||||
| Weighted average grant date fair value |
$10.01 | $8.90 | ||||||
| Weighted average risk-free interest rate |
0.67 | % | 1.0 | % | ||||
| Expected option life in years |
4.9 | 5.3 | ||||||
| Dividend yield |
1.56% - 2.00 | % | 0 | % | ||||
| Expected volatility |
32% - 34 | % | 31 - 34 | % | ||||
| ESPP purchase rights: |
||||||||
| Weighted average risk-free interest rate |
0.1 | % | 0.1 | % | ||||
| Expected option life in years |
6 months | 6 months | ||||||
| Dividend yield |
1.44% - 1.67 | % | 0 | % | ||||
| Expected volatility |
24% - 30 | % | 24% - 40 | % | ||||
- F22 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (16) | Stock-based Employee Compensation, Continued |
During the fiscal year ended June 30, 2013, we also granted performance restricted stock units (PRSUs), which contain a market condition, with the ultimate realizable number of PRSUs dependent on relative total stockholder return over a three-year period, up to a maximum amount to be issued under the award of 200% of the original grant. The weighted average grant date fair value of PRSUs granted during the year was estimated at $38.46 per PRSU using a Monte-Carlo simulation valuation model.
| (17) | Employee Retirement Plans |
We contribute to a number of employee retirement plans for the benefit of our employees. Details of the main plans are as follows:
(1) Australia - We contribute to defined contribution plans for each employee resident in Australia. All Australian employees, after serving a qualifying period, are entitled to benefits on retirement, disability or death. Employees may contribute additional funds to the plans. We contribute to the plans at the rate of 9% of the salaries of all Australian employees. Our total contributions to the plans for the years ended June 30, 2013, 2012 and 2011, were $10.3 million, $9.5 million and $8.2 million, respectively.
(2) United Kingdom - We contribute to a defined contribution plan for each permanent United Kingdom employee. All employees, after serving a three-month qualifying period, are entitled to benefit on retirement, disability or death. Employees may contribute additional funds to the plan. We contribute to the plan at the rate of 5% of the salaries of all United Kingdom employees. Our total contributions to the plan were $0.3 million, $0.3 million and $0.3 million in fiscal 2013, 2012 and 2011, respectively.
(3) United States - We sponsor a defined contribution plan available to substantially all domestic employees. Company contributions to this plan are based on a percentage of employee contributions to a maximum of 4% of the employees salary. Our total contributions to the plan were $2.9 million, $2.4 million and $2.2 million in fiscal 2013, 2012 and 2011, respectively.
(4) Switzerland - We sponsor a fixed return defined contribution fund for each permanent Swiss employee. As part of our contribution to the fund, we guarantee a fixed 2% net return on accumulated contributions per annum. We contribute to the plan at variable rates that have averaged 8% of salaries over the last three years. Our total contributions to the plan were $0.4 million, $0.4 million and $0.3 million in fiscal 2013, 2012 and 2011, respectively.
| (18) | Commitments |
We lease buildings, motor vehicles and office equipment under operating leases. We expense rental charges for operating leases on a straight-line basis over the lease term taking into account rent concessions or holidays. Rent expenses under operating leases for the years ended June 30, 2013, 2012 and 2011 were approximately $15.2 million, $13.8 million and $14.3 million, respectively. At June 30, 2013 we had the following future minimum lease payments under non-cancelable operating leases (in thousands):
| Years | Operating Leases | |||
| 2014 |
$ | 15,024 | ||
| 2015 |
11,456 | |||
| 2016 |
6,156 | |||
| 2017 |
2,990 | |||
| 2018 |
997 | |||
| Thereafter |
668 | |||
| Total minimum lease payments |
$ | 37,291 | ||
- F23 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (18) | Commitments, Continued |
Details of other commercial commitments at June 30, 2013 are as follows (in thousands):
|
In $000s |
Total |
Amount of Commitment Expiration Per Period | ||||||||||||||||||||||||||
| 2014 | 2015 | 2016 | 2017 | 2018 | Thereafter | |||||||||||||||||||||||
| Purchase Obligations |
$ | 90,827 | $ | 90,827 | $ | - | $ | - | $ | - | $ | - | $ | - | ||||||||||||||
| Guarantees* |
15,307 | 3,074 | 715 | 31 | - | 4,208 | 7,279 | |||||||||||||||||||||
| Other |
1,460 | 365 | 365 | 365 | 365 | - | - | |||||||||||||||||||||
| Total |
$ | 107,594 | $ | 94,266 | $ | 1,080 | $ | 396 | $ | 365 | $ | 4,208 | $ | 7,279 | ||||||||||||||
* The above guarantees mainly relate to requirements under contractual obligations with insurance companies transacting with our German subsidiaries and guarantees provided under our facility leasing obligations.
| (19) | Legal Actions and Contingencies |
Litigation
In the normal course of business, we are subject to routine litigation incidental to our business. While the results of this litigation cannot be predicted with certainty, we believe that their final outcome will not, individually or in aggregate, have a material adverse effect on our consolidated financial statements taken as a whole.
In February 2007, the University of Sydney (University) commenced legal action in the Federal Court of Australia against us, claiming breach of a license agreement and infringement of certain intellectual property. In October 2007, we filed a defense denying the Universitys claim, as well as a cross-claim against the University seeking an order for rectification of the contract and alleging the University violated the Australian Trade Practices Act. In May 2013, during compulsory mediation ordered by the court, we reached an agreement with the University, under which all outstanding claims were settled - see note 23 - Education, Research and Settlement Charge for further details. In June 2013, in accordance with the settlement, the Federal Court dismissed the proceedings.
In March 2013, we filed parallel legal actions in the International Trade Commission, or ITC, and in U.S. district court against Taiwanese manufacturer APEX Medical and its U.S. distributor to stop the infringement of several ResMed patents. In July 2013, the ITC entered a consent decree against the U.S. distributor, ordering that it not import or sell after import certain products we accused of infringing our patents. In July 2013, an ITC administrative law judge recommended the commission enter a similar consent decree against Apex. The ITC has not yet entered the order. The matter is ongoing as to Apex in the ITC and as to both parties in the US district court. However, we do not expect the outcome of this matter to have a material effect on our consolidated financial statements when taken as a whole.
In June 2013, we filed a lawsuit in the U.S. district court against Chinese manufacturer BMC Medical Co., Ltd and its U.S. affiliates to stop the infringement of several ResMed patents. In July 2013, we amended the district court lawsuit, and filed a parallel proceeding in the ITC. The ITC has not yet acted on our complaint, and BMC has not yet responded to the district court lawsuit. The matter is ongoing. However, we do not expect the outcome of this matter to have a material effect on our consolidated financial statements when taken as a whole.
| (20) | Fair Value Measurements |
In determining the fair value measurements of our financial assets and liabilities, we consider the principal and most advantageous market in which we transact and consider assumptions that market participants
- F24 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (20) | Fair Value Measurements, Continued |
would use when pricing the financial asset or liability. We maximize the use of observable inputs and minimize the use of unobservable inputs when measuring fair value.
The hierarchies of inputs are as follows:
| Level 1: |
Input prices quoted in an active market for identical financial assets or liabilities; | |
| Level 2: |
Inputs other than prices quoted in Level 1, such as prices quoted for similar financial assets and liabilities in active markets, prices for identical assets and liabilities in markets that are not active or other inputs that are observable or can be corroborated by observable market data; and | |
| Level 3: |
Input prices quoted that are significant to the fair value of the financial assets or liabilities which are not observable nor supported by an active market. | |
The following table summarizes our financial assets and liabilities, as at June 30, 2013 and June 30, 2012, using the valuation input hierarchy (in thousands):
| Level 1 | Level 2 | Level 3 | Total | |||||||||||||
| Balances at June 30, 2013 |
||||||||||||||||
| Foreign currency hedging instruments, net |
$ | - | $ | (7,000 | ) | $ | - | $ | (7,000 | ) | ||||||
| Business acquisition contingent consideration |
$ | - | $ | - | $ | (7,779 | ) | $ | (7,779 | ) | ||||||
| Balances at June 30, 2012 |
||||||||||||||||
| Foreign currency hedging instruments |
$ | - | $ | 14,631 | $ | - | $ | 14,631 | ||||||||
| Business acquisition contingent consideration |
$ | - | $ | - | $ | (5,024 | ) | $ | (5,024 | ) | ||||||
We determine the fair value of our financial assets and liabilities as follows:
Foreign currency options - These financial instruments are valued using third-party valuation models based on market observable inputs, including interest rate curves, on-market spot currency prices, volatilities and credit risk.
Contingent consideration - These liabilities include the fair value estimates of additional future payments that may be required for some of our previous business acquisitions based on the achievement of certain performance milestones. Each potential future payment is valued using the estimated probability of achieving each milestone, which is then discounted to present value.
The following is a reconciliation of changes in the fair value of contingent consideration during fiscal year ended June 30, 2013 (in thousands):
| Fiscal Year Ended June 30, 2013 | ||||
| Balance at the beginning of the period |
$ | (5,024 | ) | |
| Acquisition date fair value of contingent consideration |
(12,150 | ) | ||
| Changes in fair value included in operating income |
1,827 | |||
| Payments |
7,790 | |||
| Foreign currency translation adjustments |
(222 | ) | ||
| Balance at the end of the period |
$ | (7,779 | ) |
We did not have any significant non-financial assets or liabilities measured at fair value on June 30, 2013 or June 30, 2012.
- F25 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (21) | Derivative Instruments and Hedging Activities |
We transact business in various foreign currencies, including a number of major European currencies as well as the Australian and Singapore dollars. We have significant foreign currency exposure through both our Australian and Singaporean manufacturing activities, and international sales operations. We have established a foreign currency hedging program using purchased currency options and forward contracts to hedge foreign-currency-denominated financial assets, liabilities and manufacturing cash flows. The terms of such foreign currency hedging contracts generally do not exceed three years. The goal of this hedging program is to economically manage the financial impact of foreign currency exposures denominated mainly in Euros, Australian and Singapore dollars. Under this program, increases or decreases in our foreign currency denominated financial assets, liabilities, and firm commitments are partially offset by gains and losses on the hedging instruments.
We do not designate these foreign currency contracts as hedges. We have determined our hedge program to be a non-effective hedge as defined under the FASB issued authoritative guidance. All movements in the fair value of the foreign currency instruments are recorded within other income, net in our consolidated statements of income. We do not enter into financial instruments for trading or speculative purposes.
We held foreign currency instruments with notional amounts totaling $462.1 million and $334.7 million at June, 2013 and June 30, 2012, respectively, to hedge foreign currency fluctuations. These contracts mature at various dates prior to June 30, 2016.
The following table summarizes the amount and location of our derivative financial instruments as of June 30, 2013 and June 30, 2012 (in thousands):
| June 30, 2013 | June 30, 2012 | Balance Sheet Caption | ||||||||||
| Foreign currency hedging instruments |
$ | 1,350 | $ | 10,823 | Other assets - current | |||||||
| Foreign currency hedging instruments |
657 | 3,808 | Other assets-non current | |||||||||
| Foreign currency hedging instruments |
(9,007 | ) | - | Accrued expenses | ||||||||
| $ | (7,000 | ) | $ | 14,631 | ||||||||
The following table summarizes the amount and location of gains (losses) associated with our derivative financial instruments and other foreign-currency-denominated transactions for the three and twelve months ended June 30, 2013 and June 30, 2012, respectively (in thousands):
| Gain /(Loss) Recognized | Income Statement Caption | |||||||||||
| Year Ended June 30, | ||||||||||||
| 2013 | 2012 | |||||||||||
| Foreign currency hedging instruments |
$ | (21,114 | ) | $ | 18,123 | Other, net | ||||||
| Other foreign-currency-denominated transactions |
19,212 | (8,357 | ) | |||||||||
| $ | (1,902 | ) | $ | 9,766 | Other, net | |||||||
We are exposed to credit-related losses in the event of non-performance by counter parties to financial instruments. We minimize counterparty credit risk by entering into derivative transactions with major financial institutions and we do not expect material losses as a result of default by our counterparties.
| (22) | Business Combinations |
On July 20, 2012, we acquired 100% of the outstanding shares of Umbian Inc., an innovative data services technology provider, based in Nova Scotia, Canada. Umbian offers a comprehensive patient compliance management solution, which monitors continuous positive airway pressure (CPAP) devices and provides a suite of interactive follow-up services for healthcare providers. The initial purchase price was $5.6 million with an additional potential earn-out payment of up to $35.4 million based on the achievement of certain performance milestones, up to June 30, 2014, of which we have recognized a liability of $12.2 million. The
- F26 -
Table of Contents
RESMED INC. AND SUBSIDIARIES
Notes to Consolidated Financial Statements
| (22) | Business Combinations, Continued |
acquisition has been accounted for as a business combination using purchase accounting and is included in our consolidated financial statements from July 20, 2012. The acquisition is not considered a material business combination and accordingly pro forma information is not provided. The acquisition was funded through cash on-hand and we have not incurred any material acquisition related costs.
The cost of the acquisition has been allocated to the assets acquired and liabilities assumed based on estimates of their fair values at the date of acquisition. We completed the purchase price allocation during the quarter ended December 31, 2012. As part of the final purchase price allocation, we recognized an intangible asset relating to developed technology of $5.5 million and goodwill of $13.9 million. The goodwill recognized as part of this acquisition, which is not deductible for tax purposes, mainly represents the synergies that are unique to our combined businesses and the potential for new products and services to be developed in the future.
| (23) | Education, Research and Settlement Charge |
The education, research and settlement charge relates to an agreement with the University of Sydney (University) in May 2013 to pay $24.8 million to the University comprising the following:
| | $9.9 million towards the establishment of two perpetual academic chairs called the ResMed Chair of Sleep Medicine for sleep-disordered breathing with a focus on chronic disease and the ResMed Chair of Biomedical Engineering with an emphasis on bioinformatics research. |
| | $4.9 million in equal installments, over five years, to be used by the University to support research in the fields of sleep medicine and biomedical engineering. |
| | $10.0 million in settlement of proceedings between the parties, including $1.25 million for the release from the proceedings and $8.75 million for prior period royalties payable under the license agreement. |
We have expensed the full amount of the charge, in our operating income, in fiscal year 2013.
- F27 -
Table of Contents
SCHEDULE II
RESMED INC. AND SUBSIDIARIES
VALUATION AND QUALIFYING ACCOUNTS AND RESERVES
YEARS ENDED JUNE 30, 2013, 2012 AND 2011
(in thousands)
| Balance at Beginning of Period |
Charged to costs and expenses |
Other (deductions) |
Balance at end of period |
|||||||||||||
| Year ended June 30, 2013 |
||||||||||||||||
| Applied against asset account |
||||||||||||||||
| Allowance for doubtful accounts |
$ | 7,313 | 5,939 | (3,339 | ) | $ | 9,912 | |||||||||
| Year ended June 30, 2012 |
||||||||||||||||
| Applied against asset account |
||||||||||||||||
| Allowance for doubtful accounts |
$ | 11,476 | 2,652 | (6,815 | ) | $ | 7,313 | |||||||||
| Year ended June 30, 2011 |
||||||||||||||||
| Applied against asset account |
||||||||||||||||
| Allowance for doubtful accounts |
$ | 7,826 | 5,210 | (1,560 | ) | $ | 11,476 | |||||||||
See accompanying report of independent registered public accounting firm.
Table of Contents
SIGNATURES
Under the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has caused this report to be signed on its behalf by the authorized persons below.
DATED August 12, 2013
ResMed Inc.
| /S/ MICHAEL J. FARRELL |
| MICHAEL J. FARRELL |
| Chief executive officer (Principal Executive Officer) |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the Registrant and in the capacities and on the dates indicated.
| SIGNATURE | TITLE | DATE | ||
| /S/ MICHAEL J. FARRELL Michael J. Farrell |
Chief executive officer and director (Principal Executive Officer) | August 12, 2013 | ||
| /S/ BRETT A. SANDERCOCK Brett A. Sandercock |
Chief financial officer (Principal Financial Officer and Principal Accounting Officer) |
August 12, 2013 | ||
| /S/ PETER C. FARRELL Peter C. Farrell |
Executive chairman |
August 12, 2013 | ||
| /S/ CHRISTOPHER G. ROBERTS Christopher G. Roberts |
Director |
August 12, 2013 | ||
| /S/ MICHAEL A. QUINN Michael A. Quinn |
Director |
August 12, 2013 | ||
| /S/ GARY W. PACE Gary W. Pace |
Director |
August 12, 2013 | ||
| /S/ RICHARD SULPIZIO Richard Sulpizio |
Director |
August 12, 2013 | ||
| /S/ RON TAYLOR Ron Taylor |
Director |
August 12, 2013 | ||
| /S/ JOHN P. WAREHAM John P. Wareham |
Director |
August 12, 2013 | ||
S-1
Table of Contents
RESMED INC. AND SUBSIDIARIES
EXHIBIT INDEX
The following documents are filed as part of this report:
| (a) | Consolidated Financial Statements and Schedules The index to our consolidated financial statements and schedules are set forth in the Index to Consolidated Financial Statements under Item 8 of this report. | |
| (b) | Exhibit Lists | |
| 3.1 | First Restated Certificate of Incorporation of Registrant, as amended. (5) | |
| 3.2 | Fifth Amended and Restated Bylaws of ResMed Inc. (17) | |
| 4.1 | Form of certificate evidencing shares of Common Stock. (1) | |
| 10.1 | Licensing Agreement between the University of Sydney and ResMed Ltd dated May 17, 1991, as amended. (1) | |
| 10.2* | ResMed Inc. 2006 Incentive Award Plan. (6) | |
| 10.3* | Amendment No. 1 to the ResMed Inc. 2006 Incentive Award Plan. (3) | |
| 10.4* | 2006 Grant agreement for Board of Directors. (3) | |
| 10.5* | 2006 Grant agreement for Executive Officers. (5) | |
| 10.6* | 2006 Grant agreement for Australian Executive Officers. (5) | |
| 10.7* | Form of Executive Agreement. (4) | |
| 10.8* | Amended and Restated 2006 Incentive Award Plan dated November 20, 2008. (7) | |
| 10.9 | Departure of Directors or Certain Officers dated December 12, 2008. (8) | |
| 10.10 | Approval of new share repurchase program dated May 29, 2009. (9) | |
| 10.11 | Form of Indemnification Agreements for our directors and officers. (10) | |
| 10.12 | Form of Access Agreement for directors. (10) | |
| 10.13* | Updated Form of Executive Agreement. (2)(16) | |
| 10.14 | ResMed Inc. 2009 Incentive Award Plan. (11) | |
| 10.15 | ResMed Inc. 2009 Employee Stock Purchase Plan. (11) | |
| 10.16 | Amendment No. 1 to the ResMed Inc. 2009 Employee Stock Purchase Plan (18) | |
| 10.17 | Form of Restricted Stock Award Agreement. (11) | |
| 10.18 | ResMed Inc. Deferred Compensation Plan. (12) | |
| 10.19 | Credit Agreement, dated February 10, 2011, by and between ResMed Inc. and the lenders, including Union Bank, N.A., HSBC Bank USA, National Association, Commonwealth Bank of Australia and Wells Fargo Bank, N.A. (13) | |
| 10.20 | First Amendment to Credit Agreement, dated January 25, 2012, by and between ResMed Inc. and the lenders, including Union Bank, N.A., HSBC Bank USA, National Association, Commonwealth Bank of Australia and Wells Fargo Bank, N.A. (15) | |
| 10.21 | Pledge and Security Agreement, dated as of February 10, 2011, by and between ResMed Inc., as Pledgor, and Union Bank, N.A., as Administrative Agent. (13) | |
Table of Contents
| 10.22 | Unconditional Guaranty entered into as of February 10, 2011, by each of ResMed Corp., ResMed Assembly US Inc. and ResMed Motor Technologies Inc., in favor of Union Bank, N.A., as Administrative Agent. (13) | |
| 10.23 | Form of Restricted Stock Unit Award Agreement for Executive Officers. (14) | |
| 10.24 | Form of Restricted Stock Unit Award Agreement for Directors. (14) | |
| 10.25 | Form of Stock Option Grant for Executive Officers. (14) | |
| 10.26 | Form of Stock Option Grant for Directors. (14) | |
| 10.27 | Form of Performance-Based Restricted Stock Unit Award Agreement for Executive Officers.(19) | |
| 21.1 | Subsidiaries of the Registrant. (20) | |
| 23.1 | Consent of Independent Registered Public Accounting Firm. (20) | |
| 31.1 | Certification of Chief Executive Officer Pursuant to Section 302 of Sarbanes-Oxley Act of 2002. (20) | |
| 31.2 | Certification of Chief Financial Officer Pursuant to Section 302 of Sarbanes-Oxley Act of 2002. (20) | |
| 32.1 | Certification of Chief Executive Officer and Chief Financial Officer pursuant to Section 906 of the Sarbanes-Oxley Act of 2002. (20) | |
| 101 | The following materials from ResMed Inc.s Annual Report on Form 10-K for the fiscal year ended June 30, 2013 formatted in Extensible Business Reporting Language (XBRL): (i) the Consolidated Balance Sheets, (ii) the Consolidated Statements of Income, (iii) the Consolidated Statements of Stockholders Equity and Comprehensive Income, (iv) the Consolidated and Statements of Cash Flows and (v) related notes. | |
| * | Management contract or compensatory plan or arrangement |
| (1) | Incorporated by reference to the Registrants Registration Statement on Form S-1 (No. 33-91094) declared effective on June 1, 1995. |
| (2) | Incorporated by reference to the Registrants Report on Form 10-K for the year ended June 30, 2009. |
| (3) | Incorporated by reference to the Registrants Report on Form 10-Q for the quarter ended December 31, 2006. |
| (4) | Incorporated by reference to the Registrants Report on Form 8-K filed on July 13, 2007. |
| (5) | Incorporated by reference to the Registrants Report on Form 10-K for the year ended June 30, 2007. |
| (6) | Incorporated by reference to the Registrants Report on Form 8-K filed on November 15, 2006. |
| (7) | Incorporated by reference to Appendix A of the Registrants Definitive Proxy Statement filed on October 15, 2008. |
| (8) | Incorporated by reference to the Registrants Report on Form 8-K filed on December 15, 2008. |
| (9) | Incorporated by reference to the Registrants Report on Form 8-K filed on June 4, 2009. |
| (10) | Incorporated by reference to the Registrants Report on Form 8-K filed on June 24, 2009. |
| (11) | Incorporated by reference to the Registrants Report on Form 8-K filed on November 23, 2009. |
| (12) | Incorporated by reference to the Registrants Report on Form 8-K filed on May 25, 2010. |
| (13) | Incorporated by reference to the Registrants Report on Form 8-K filed on February 14, 2011. |
| (14) | Incorporated by reference to the Registrants Report on Form 10-Q for the quarter ended September 30, 2011. |
| (15) | Incorporated by reference to the Registrants Report on Form 8-K filed on January 26, 2012. |
| (16) | Incorporated by reference to the Registrants Report on Form 8-K filed on July 2, 2012. |
| (17) | Incorporated by reference to the Registrants Report on Form 8-K/A filed on September 17, 2012. |
| (18) | Incorporated by reference to Appendix A of the Registrants Definitive Proxy Statement filed on October 4, 2012. |
| (19) | Incorporated by reference to the Registrants Report on Form 8-K filed on November 21, 2012. |
| (20) | Filed with this report. |